Fri, Jul 11, 2025
Volume 5, Issue 1 (Winter 2019)
Iran J Neurosurg 2019, 5(1): 21-30 |
Back to browse issues page
Download citation:
BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:



BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:
Abrishamkar S, Sadeghi M B, Tabesh H, Khani H. Evaluating Demographic and Clinical Characteristics and Type of Hydrocephalus Before and After Surgical Interventions in Patients With Intraventricular Brain Tumors. Iran J Neurosurg 2019; 5 (1) :21-30
URL: http://irjns.org/article-1-166-en.html
URL: http://irjns.org/article-1-166-en.html
1- Professor, Department on Neurosurgery, Medical School, Isfahan University of Medical Sciences, Isfahan, Iran
2- Resident of Neurosurgery, Medical School, Isfahan University of Medical Sciences, Isfahan, Iran ,dr.mb_sadeghi@yahoo.com
3- Associate Professor, Department of Neurosurgery, Medical School, Isfahan University of Medical Sciences, Isfahan, Iran
4- Resident of Neurosurgery, Medical School, Isfahan University of Medical Sciences, Isfahan, Iran
2- Resident of Neurosurgery, Medical School, Isfahan University of Medical Sciences, Isfahan, Iran ,
3- Associate Professor, Department of Neurosurgery, Medical School, Isfahan University of Medical Sciences, Isfahan, Iran
4- Resident of Neurosurgery, Medical School, Isfahan University of Medical Sciences, Isfahan, Iran
Full Text [PDF 745 kb]
(1498 Downloads)
| Abstract (HTML) (4515 Views)
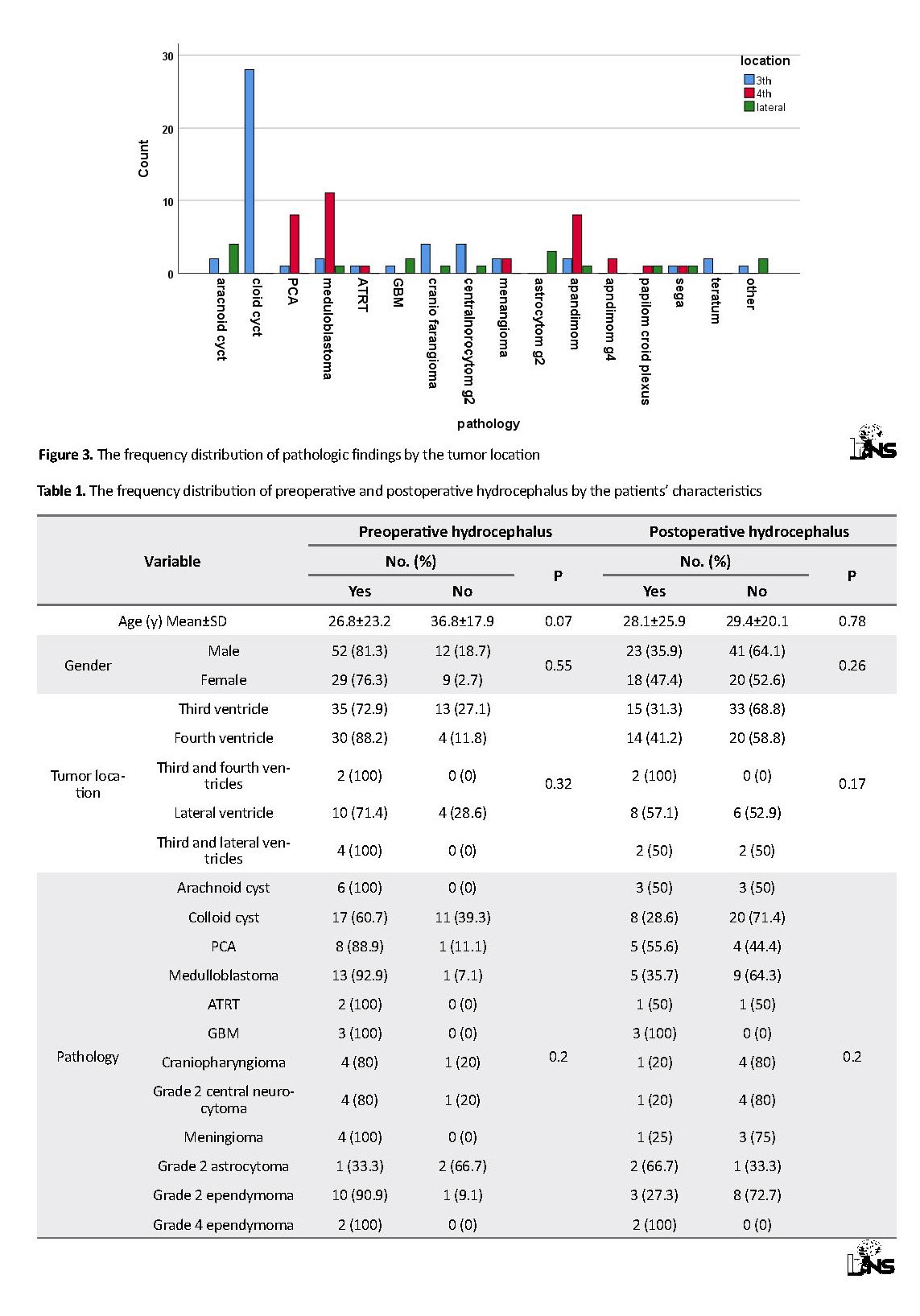
Evaluating the tumor pathology and location also found colloidal cyst with a prevalence of 28 (100%) to be the most prevalent tumor in the third ventricle, medulloblastoma with a prevalence of 11 (78.6%) to be the most prevalent tumor in the fourth ventricle and astrocytoma with a prevalence of 3 (100%) to be the most prevalent in the lateral ventricles. The tumor pathology was significantly different in terms of its location (P<0.001) (figure 3).
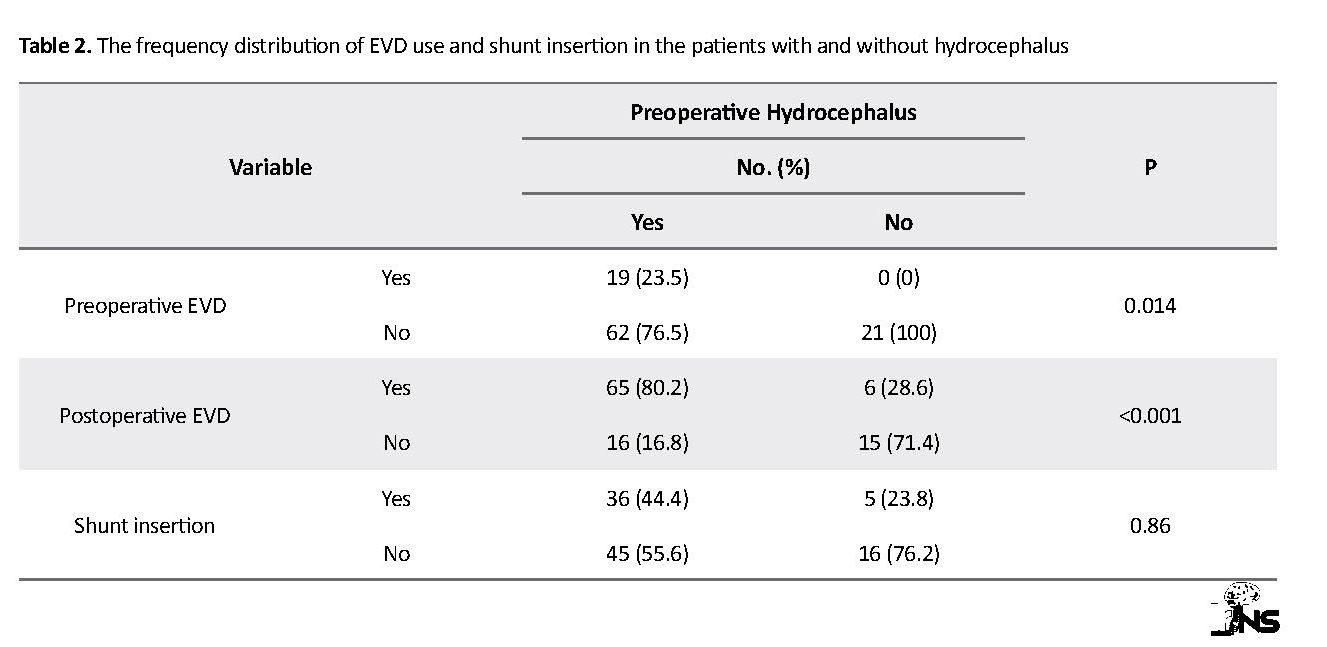
Comparing the results of the 102 patients showed preoperative hydrocephalus in 81 (79.4%) patients, 41 (40.2%) of whom received a shunt. Hydrocephalus was observed in 35.3% (n=36) of the patients before and after the surgery, whereas it was not diagnosed in 15.7% (n=16) before the operation. Only postoperative, rather than preoperative, hydrocephalus was diagnosed in 4.9% (n=5) of the patients, whereas preoperative, rather than postoperative, hydrocephalus was diagnosed in 44.1% (n=45) of the patients.
Table 1 presents the frequency distribution of hydrocephalus before and after the surgery by age, gender, tumor location and tumor pathology. According to table 1 and the t-test results, despite the higher mean age of the patients with hydrocephalus, no significant differences were observed between the patients with and without hydrocephalus either before or after the surgery (P=0.78).
The prevalence of preoperative hydrocephalus was 81.3% in the men and 76.3% in the women (P=0.55), and that of postoperative hydrocephalus was 35.9% in the men and 47.4% in the women (P=0.26), suggesting no significant differences between the two genders. In terms of the tumor location, the prevalence of preoperative hydrocephalus was higher in the third and fourth ventricles and the third and lateral ventricles, although the difference was insignificant (P=0.32). In terms of the operative site, the third and fourth ventricles were the most prevalent sites of the tumor, although post-operative hydrocephalus was not significantly and statistically associated with the tumor location (P=0.17).
Preoperative hydrocephalus was pathologically observed in all the cases of arachnoid cysts, ATRT, GBM, meningium, grade 4 ependymoma and teratoma (P=0.2). Postoperative hydrocephalus was also reported in all the cases of GBM, grade 4 ependymoma and teratoma (P=0.2). The tumor pathology, however, did not show any significant differences in the incidence of hydrocephalus between before and after the surgery.
An EVD was used to improve hydrocephalus in 19 (18.6%) cases before the surgery and 71 (69.6%) afterwards. A total of 23.5% of the patients with hydrocephalus received an EVD, and significant differences were observed in the use of EVD before the operation (P=0.014). The frequency of EVD in operation was also 80.2% in the patients with hydrocephalus and 28.6% in those without hydrocephalus. Moreover, statistically-significant relationships were found between the use of postoperative EVD and the absence of preoperative hydrocephalus (P<0.001). Out of the 102 patients undergoing the surgery, 41 (40.2%) were treated with a shunt. The frequency of shunt insertion was 44.4% in the patients with hydrocephalus and 23.8% in those without hydrocephalus, suggesting no significant differences in shunt insertion between the two groups (P>0.066). Table 2 presents the frequency distribution of EVD use and shunt insertion in the patients with and without hydrocephalus.
According to table 3, the frequency of EVD was 20.8% in third ventricular tumors, 17.6% in fourth ventricular tumors, 100% in third and fourth ventricular tumors and 25% in third and lateral ventricular tumors, and the preoperative use of EVD was significantly different by the tumor location (0.016). The frequency of EVD usage was also significantly different by the tumor location, with 54.2% in third ventricular tumors, 85.3% in fourth ventricular tumors, 71.4% in lateral ventricular tumors and 100% in third ventricular tumors plus fourth ventricle and third+lateral ventricles (P=0.017). No significant differences were also observed in shunt insertion by the tumor location (P=0.17).
According to table 4 and the tumor pathology, no significant differences were observed in the distribution of EVD between before and after the operation.
4. Discussion
As a serious complication in patients with intraventricular tumors, hydrocephalus can exacerbate the disease symptoms, and cause death if not treated in a timely manner. Preventing hydrocephalus in patients undergoing a cerebral surgery is therefore considered a priority in postoperative care. Despite the importance of this complication, no comprehensive studies have yet been conducted in Iran on the incidence of hydrocephalus and the demographic and clinical features of the patients. The present study was conducted to determine demographic and clinical features and type of hydrocephalus therapeutic interventions before and after the surgery in patients with intraventricular brain tumors.
The present findings suggested that colloid cyst, medulloblastoma and grade 2 ependymoma were the most prevalent tumor in the patients with intraventricular tumors. In 2018, Pereira et al. also found colloid cysts to be the most prevalent intraventricular tumors [7]. The incidence and prevalence of these tumors have significantly increased in recent years due to different causes, including the increased risk factors for brain tumors, increased exposure to risk factors, advances in diagnostic and therapeutic facilities and new classifications of brain tumors [8].
Investigating demographic characteristics found below 10 years with a frequency of 31.4% to be the most prevalent age in the patients with intraventricular tumors. These tumors were also observed in over 60% of the males. Moreover, June et al. found the prevalence of intraventricular tumors to be higher in younger ages, the mean age of the patients to be 36 years and over two thirds of them to be male [9]. According to the results of the present study, the tumor pathology was not significantly different between the men and women, although it was significantly different among the different age groups. Contro et al. found the tumor pathology to be significantly different among the age groups of the patients, and the patients with SEGA and ATRT to be the youngest [10], which is consistent with the present study.
Investigating the tumor location and its associations with demographic characteristics found the third ventricle with a frequency of 47.1% to be the most prevalent site of intraventricular tumors. Furthermore, the tumor pathology was significantly related to the tumor location in a way that all the colloidal cysts lay in the third ventricle and 78.6% of the medulloblastomas lay in the fourth ventricle. Waldron et al. reported significant associations between the type and location of the tumor [8]. They found the prevalence of hydrocephalus to be 79.4% in the patients undergoing intraventricular tumor surgeries.
A hydrocephalic estimation in 2016 by Peyellow et al. showed that shunt insertion was more prevalent in patients younger than two years old, moderate-to-severe preoperative hydrocephalus, brain metastases and pathology of medulloblastoma and ependymoma compared to that in other cases [11]. In addition, a study by Guive et al. in 2014 on the endoscopic treatment of 53 patients with third-ventricle non-hydrocephalic colloid cysts found only two patients to require treatments for post-operative hydrocephalus [12]. A study conducted by Jay Talin et al. in 2015 on evaluating and treating hydrocephalus in children with posterior fossa tumors reported young age, preoperative hydrocephalus, metastasis, middle-line tumors, tumor pathology, subtotal tumor resection and EVD use in continuous post-operative hydrocephalic outbreaks [13].
A study conducted by Symss et al. in India in 2011 on 78 patients with colloid cysts found the prevalence of preoperative hydrocephalus to be 83.4%, which is consistent with the present research, although they found shunt insertion to be eventually required in four of the patients, which is less than the corresponding figure in the present study [14]. A study conducted by Cotton et al. in 2002 on 105 patients with colloid cysts reported shunt insertion requirements in 23 of the patients, which is consistent with the shunt insertion requirements in the present study [15]. The difference in the incidence of shunt insertion can be explained by the differences in the surgical technique used, the amount of surgical manipulation and intraoperative bleeding and the ability to completely remove colloid cysts without tearing the content of the cyst into the ventricle.
Using interhemispheric approaches and younger ages were found correlated with postoperative hydrocephalus [8], although it was not confirmed in the majority of similar studies. Furthermore, Philippe Kapley et al. reported different degrees of hydrocephalus in 29 patients with intraventricular tumors. They found that ventriculocystotomy alone was associated low success rates, and that it can eliminate the need for a shunt in the following 5 years in 83% of the cases if it is used in combination with endoscopic cisternotomy [9].
The present study found all the intra-cerebroventricular arachnoid cases to be hydrocephalus prior to the surgery, and half of the patients to need shunt insertion after the surgery. It is worth noting the open type of the surgical technique used and only performing the resection of the walls of the cysts. A study conducted by Eitan et al. in 2016 found 18% of patients diagnosed with intraventricular ependymoma in the fourth ventricle to need shunt insertion after the tumor resection [10], whereas the present study found this figure to be 27.3% in grade 2 ependymoma and 100% in grade 4.
5. Conclusion
Some features, including the pathology and location of the tumor, appeared effective in the subsequent hydrocephalic survival both before and after the operation. If these findings are confirmed and given these factors, therapeutic measures such as EVD and shunt insertion can be considered for preventing and treating postoperative hydrocephalus. Given the limitations of the present study, including a small sample, further research is recommended that be conducted using larger samples.
Ethical Considerations
Compliance with ethical guidelines
The present study was approved by the Ethics Committee of Isfahan University of Medical Sciences. Informed consent was obtained from all the participants.
Funding
This study was supported by Isfahan University of Medical Sciences. The study design was registered at the research faculty of the School of Medicine, Isfahan University of Medical Sciences (Code: 395112) The present article was partly extracted from a thesis of Mohammad Bagher Sadeghi on Neurosurgery.
Authors contributions
Study idea, design, interpretation and writing the paper: Saeid Abrishamkar; Data analysis and interpretation, preparing and writing the paper, submitting the manuscript and corresponding with the reviewers: Mohammad Bagher Sadeghi.
Conflict of interest
The authors declared no conflicts of interest regarding the publication of the present article.
Acknowledgements
The authors would like to express their gratitude to the authorities of School of Medicine, Isfahan University of Medical Sciences, for their efforts.
References
Jemal A, Siegel R, Xu J, Ward E. Cancer statistics, 2010. CA: A Cancer Journal for Clinicians. 2010; 60(5):277-300. [DOI:10.3322/caac.20073] [PMID]
Ohgaki H, Kleihues P. Population-based studies on incidence, survival rates, and genetic alterations in astrocytic and oligodendroglial gliomas. Journal of Neuropathology & Experimental Neurology. 2005; 64(6):479-89. [DOI:10.1093/jnen/64.6.479] [PMID]
Ge L, Hoa NT, Cornforth AN, Bota DA, Mai A, Kim DI, et al. Glioma big potassium channel expression in human cancers and possible T cell epitopes for their immuneotherapy. The Journal of Immunology. 2012; 189(5):2625-34. [DOI:10.4049/jimmunol.1102965] [PMID] [PMCID]
Burger PC, Schelthauer BW, Vogel FS,editors. Surgical pathology of the nervous system and its covering. 3rd ed. New York: Churchill Livingstone; 1991.
Brown PD, Decker PA, Rummans TA, Clark MM, Frost MH, Ballman KV, et al. A prospective study of quality of life in adults with newly diagnosed high-grade gliomas: Comparison of patient and caregiver ratings of quality of life. American Journal of Clinical Oncology. 2008; 31(2):163-8. [DOI:10.1097/COC.0b013e318149f1d3] [PMID]
Aaronson NK. Quality of life: What is it? How should it be measured? Oncol (Williston Park). 1988; 2(5):69-76.
Pereira BJ, de Almeida AN, Paiva WS, de Aguiar PH, Teixeira MJ, Marie SK. Natural history of intraventricular meningiomas: systematic review. Neurosurgical review. 2018; 15:1-11. [DOI:10.1007/s10143-018-1019-0]
Waldron JS, Tihan T. Epidemiology and pathology of intraventricular tumors. Neurosurgery Clinics. 2003; 14(4):469-82. [DOI:10.1016/S1042-3680(03)00060-3]
Brandsma D, van den Bent MJ. Molecular targeted therapies and chemotherapy in malignant gliomas. Current Opinion in Oncology. 2007; 19(6):598-605. [DOI:10.1097/CCO.0b013e3282f0313b] [PMID]
Jie NJ, Teo KA, Shabbir A, Yeo TT. Widespread intra-abdominal carcinomatosis from a rhabdoid meningioma after placement of a ventriculoperitoneal shunt: A case report and review of the literature. Asian journal of neurosurgery. 2018; 13(1):176-83. [DOI:10.4103/1793-5482.181128] [PMID] [PMCID]
Contro E, De Musso F, Pilu G, Ghi T. Intracranial Hemorrhage, Cysts, Tumors, and Destructive Lesions. In: Obstetric Imaging: Fetal Diagnosis and Care (2nd Edition) 2018 : 204-212. [DOI:10.1016/B978-0-323-44548-1.00040-1] [PMID] [PMCID]
Paul Foreman, m.D..Samuel mcclugage III, m.D., Robert naFtel, m.D. et al. Validation and modifcation of a predictive model of postresection hydrocephalus in pediatric patients with posterior fossa tumors. Journal of Neurosurgery: Pediatrics. 2013. 12(3):220-6. [DOI:10.3171/2013.5.PEDS1371] [PMID]
Guive Sharifi, Mehrdad Hosseinzadeh Bakhtevari, Omidvar Rezaei, Reza Jabbari: Endoscopic Surgery for Third Ventricular Colloid Cysts in the Absence of Hydrocephalus: a feasibility study. International Clinical Neuroscience Journal. 2014; 1(2):73-7.
Chih-Ta Lin & Jay K. Riva-Cambrin: Management of posterior fossa tumors and hydrocephalus in children: A review. Child's Nervous System. 2015; 31(10):1781-9. [DOI:10.1007/s00381-015-2781-8] [PMID]
Chih-Ta Lin & Jay K. Riva-Cambrin: Management of posterior fossa tumors and hydrocephalus in children: A review. Child's Nervous System. 2015; 31(10):1781-9. [DOI:10.1007/s00381-015-2781-8] [PMID]
Full Text: (1595 Views)
1. Introduction
As fearsome diseases, brain tumors [1], especially malignant ones, annually afflict about 10-15 in every 100,000 individuals. These tumors include primary brain tumors and secondary or metastatic brain tumors caused by systemic malignancies [2]. With an annual incidence of 6 per 100,000, gliomas are the most prevalent primary brain tumors arising from the glial tissue [2]. Moreover, intraventricular tumors are benign or malignant lesions emerging as intra-axial or extra-axial tumors originating from ventricles or adjacent ventricles [3]. Intraventricular tumors account for below 1% of intracranial lesions, most of which are benign and slow growing [4]. Primary intraventricular tumors originate from ventricles and secondary intraventricular tumors from the structures surrounding ventricles and invade them [4]. The widely-varying symptoms of intracerebral-ventricular tumors are caused by the blockage of the csf pathway or pressure on adjacent structures [4].
Hydrocephalus disrupts the formation and flow or absorption of cerebrospinal fluid, and increases the volume occupied by this fluid in the central nervous system. Acute hydrocephalus can develop in a matter of days or weeks, and the chronic type within months or years. Furthermore, complications such as brain atrophy and local degenerative damage can abnormally increase cerebrospinal fluid in the central nervous system [1], which increases the intracranial pressure. This complication can also be associated with increased resistance to the cerebrospinal fluid flow and increased venous sinus pressure.
The trans-ventricular absorption of cerebrospinal fluid and absorption through the channels of nerve roots constitute the compensatory mechanisms. The temporal and frontal branches are first symmetrically enlarged, which causes the corpus callosum to rise, stretch and break in the septum pellucidum, thin the brain coating and enlarge the third ventricle downward into the pituitary cavity [1]. The fluid is then absorbed by the other channels and exits the skull through the intravenous system of the brain’s surface.
Abnormal accumulation of cerebrospinal fluid can cause the blockage of the extraction channel of cerebrospinal fluid, blockage of the cerebrospinal fluid pathway around the brain and obstruction of the absorption channels of cerebrospinal fluid [1]. It is worth noting that increased intraventricular pressure enlarges them and causes the emergence of hydrocephalic symptoms [5]. Moreover, the tumor presence in the ventricle can increase the intraventricular pressure and develop hydrocephalus. Given the high incidence of brain tumors, hydrocephalic symptoms should be monitored through imaging to rule out intraventricular tumors [6]. Hydrocephalus in intraventricular tumors has been addressed in literature; nevertheless, no comprehensive studies have yet been conducted to investigate the prevalence of hydrocephalus by pathology before and after surgeries in patients with intraventricular tumors or to evaluate and manage hydrocephalic treatments.
The present study was therefore conducted to determine demographic and clinical features and type of hydrocephalus therapeutic interventions before and after surgeries in patients with intraventricular tumors.
2. Methods and Materials/Patients
The present cross-sectional study was conducted on a population comprising patients with brain tumors, who had undergone surgeries in Al-Zahra Hospital, Kashani Hospital and Imam Hossein Hospital in Isfahan, Iran in 2013-2018. The eligible candidates comprised patients with intraventricular tumors undergoing surgeries without a history of surgery, chemotherapy and radiotherapy and consenting to participation in the study. The exclusion criteria consisted of failure to follow-up after the operation, death of the patient during or after the operation and prior to evaluate hemorrhagic or hydrocephalus effects.
The sample size required for estimating the incidence of hydrocephalus in intraventricular tumors was calculated as 102 with a confidence interval of 95% and a test power of 80%. Given a lack of similar studies, the hydrocephalus prevalence was considered 0.5 in the patients undergoing the extraction of brain tumors, and an acceptable error threshold of 0.1 was included.
The present study was approved by the Ethics Committee of Isfahan University of Medical Sciences, Isfahan, Iran. A total of 102 patients whose intraventricular tumors had been diagnosed by MRI and had undergone a surgery in 2013-18 were selected and included in the study. All the patients underwent an open surgery or endoscopy of intraventricular tumors after their admission.
The patients with acute hydrocephalus who experienced a sudden decrease in acute consciousness urgently underwent EVD insertion and resection of the tumor in the fourth ventricle. The patients with abrupt acute hydrocephalus and acute hydrocephalus and lateral or third ventricular tumors underwent a tumor resection surgery or, if possible, a tumor discharge and EVD insertion under appropriate conditions. The other patients with intraventricular tumors and a GCS score of 15 underwent a tumor-discharge surgery and an EVD in case of increased intracranial pressure or severe hydrocephalus. After controlling edema and inflammation in the patients requiring an EVD, the EVD clamp was heightened. If their condition remained stable, the EVD was removed. A shunt was applied in case of EVD dependence and no reductions in the ventricular size.
The other patients with no EVD were carefully monitored from the beginning at the ICU, and in cases of having a low level of consciousness or hydrocephalus, they underwent EVD insertion and, if necessary, shunt insertion. CT scans of all the patients performed in the morning after the surgery showed no acute nerve symptoms, and their ventricular size was compared with their preoperative one. Depending on the patient’s condition, a CT scan was performed one day to one month after the surgery to compare the postoperative with the preoperative ventricular size.
Some of the patients also serially underwent a CT scan. The bicaudate ratio was used to determine the hydrocephalus level. In case of decreases in the level of consciousness or acute nerve symptoms, brain CT scans were immediately performed to obtain the pathologic outcomes of the patients. After undergoing the surgery, the patients were asked to submit the results of their preoperative MRI and postoperative CT scan to be included in the study. The data were analysed in SPSS-25 using the Chi-square test, the t-test and the Mann-Whitney U test.
3. Results
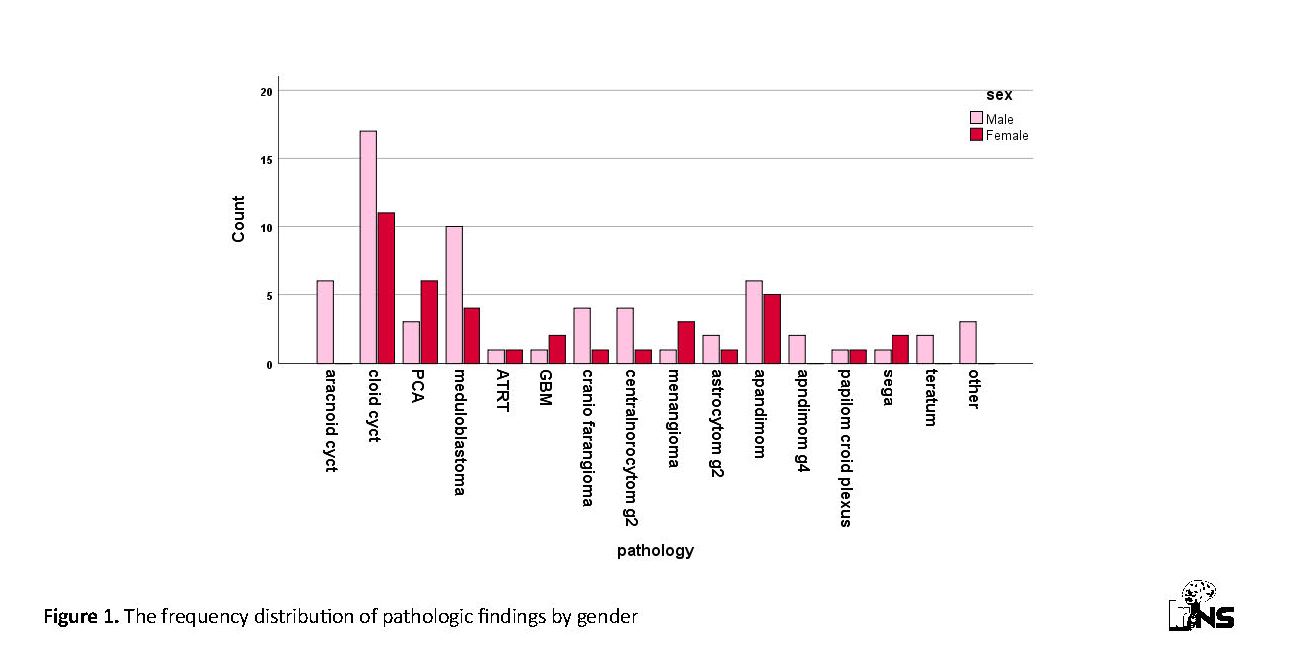
The present study was conducted on 102 patients, including 64 (62.7%) males and 38 (37.3%) females, with a mean age of 28.9±2.23 years. The tumor lay in the third ventricle in 47.1% (n=48) of the patients, the fourth ventricle in 33.3% (n=34), the lateral ventricle in 13.7% (n=14), the third and fourth ventricles in 2% (n=2) and lateral and third ventricles in 3.9% (n=4). The obtained pathologic results suggested that colloid cysts in 28 cases, moduloblastoma in 14 and grade 2 ependymoma in 11 were the most prevalent types of the tumor. Comparing the results of the tumor pathology by age and gender according to figure 1 showed that GBM, meningium and SEGA were more prevalent in the women than in the men, choroid plexus papilloma was as prevalent in the men as in the women and the prevalence of the remaining cases was higher in the men. Significant tumor-related pathological abnormalities were also observed in both genders (P=0.26). Moreover, the tumor pathology was significant in terms of age in a way that ATRT and meningioma were the most prevalent in the youngest and oldest of the patients, respectively (P<0.001) (Figure 2).
As fearsome diseases, brain tumors [1], especially malignant ones, annually afflict about 10-15 in every 100,000 individuals. These tumors include primary brain tumors and secondary or metastatic brain tumors caused by systemic malignancies [2]. With an annual incidence of 6 per 100,000, gliomas are the most prevalent primary brain tumors arising from the glial tissue [2]. Moreover, intraventricular tumors are benign or malignant lesions emerging as intra-axial or extra-axial tumors originating from ventricles or adjacent ventricles [3]. Intraventricular tumors account for below 1% of intracranial lesions, most of which are benign and slow growing [4]. Primary intraventricular tumors originate from ventricles and secondary intraventricular tumors from the structures surrounding ventricles and invade them [4]. The widely-varying symptoms of intracerebral-ventricular tumors are caused by the blockage of the csf pathway or pressure on adjacent structures [4].
Hydrocephalus disrupts the formation and flow or absorption of cerebrospinal fluid, and increases the volume occupied by this fluid in the central nervous system. Acute hydrocephalus can develop in a matter of days or weeks, and the chronic type within months or years. Furthermore, complications such as brain atrophy and local degenerative damage can abnormally increase cerebrospinal fluid in the central nervous system [1], which increases the intracranial pressure. This complication can also be associated with increased resistance to the cerebrospinal fluid flow and increased venous sinus pressure.
The trans-ventricular absorption of cerebrospinal fluid and absorption through the channels of nerve roots constitute the compensatory mechanisms. The temporal and frontal branches are first symmetrically enlarged, which causes the corpus callosum to rise, stretch and break in the septum pellucidum, thin the brain coating and enlarge the third ventricle downward into the pituitary cavity [1]. The fluid is then absorbed by the other channels and exits the skull through the intravenous system of the brain’s surface.
Abnormal accumulation of cerebrospinal fluid can cause the blockage of the extraction channel of cerebrospinal fluid, blockage of the cerebrospinal fluid pathway around the brain and obstruction of the absorption channels of cerebrospinal fluid [1]. It is worth noting that increased intraventricular pressure enlarges them and causes the emergence of hydrocephalic symptoms [5]. Moreover, the tumor presence in the ventricle can increase the intraventricular pressure and develop hydrocephalus. Given the high incidence of brain tumors, hydrocephalic symptoms should be monitored through imaging to rule out intraventricular tumors [6]. Hydrocephalus in intraventricular tumors has been addressed in literature; nevertheless, no comprehensive studies have yet been conducted to investigate the prevalence of hydrocephalus by pathology before and after surgeries in patients with intraventricular tumors or to evaluate and manage hydrocephalic treatments.
The present study was therefore conducted to determine demographic and clinical features and type of hydrocephalus therapeutic interventions before and after surgeries in patients with intraventricular tumors.
2. Methods and Materials/Patients
The present cross-sectional study was conducted on a population comprising patients with brain tumors, who had undergone surgeries in Al-Zahra Hospital, Kashani Hospital and Imam Hossein Hospital in Isfahan, Iran in 2013-2018. The eligible candidates comprised patients with intraventricular tumors undergoing surgeries without a history of surgery, chemotherapy and radiotherapy and consenting to participation in the study. The exclusion criteria consisted of failure to follow-up after the operation, death of the patient during or after the operation and prior to evaluate hemorrhagic or hydrocephalus effects.
The sample size required for estimating the incidence of hydrocephalus in intraventricular tumors was calculated as 102 with a confidence interval of 95% and a test power of 80%. Given a lack of similar studies, the hydrocephalus prevalence was considered 0.5 in the patients undergoing the extraction of brain tumors, and an acceptable error threshold of 0.1 was included.
The present study was approved by the Ethics Committee of Isfahan University of Medical Sciences, Isfahan, Iran. A total of 102 patients whose intraventricular tumors had been diagnosed by MRI and had undergone a surgery in 2013-18 were selected and included in the study. All the patients underwent an open surgery or endoscopy of intraventricular tumors after their admission.
The patients with acute hydrocephalus who experienced a sudden decrease in acute consciousness urgently underwent EVD insertion and resection of the tumor in the fourth ventricle. The patients with abrupt acute hydrocephalus and acute hydrocephalus and lateral or third ventricular tumors underwent a tumor resection surgery or, if possible, a tumor discharge and EVD insertion under appropriate conditions. The other patients with intraventricular tumors and a GCS score of 15 underwent a tumor-discharge surgery and an EVD in case of increased intracranial pressure or severe hydrocephalus. After controlling edema and inflammation in the patients requiring an EVD, the EVD clamp was heightened. If their condition remained stable, the EVD was removed. A shunt was applied in case of EVD dependence and no reductions in the ventricular size.
The other patients with no EVD were carefully monitored from the beginning at the ICU, and in cases of having a low level of consciousness or hydrocephalus, they underwent EVD insertion and, if necessary, shunt insertion. CT scans of all the patients performed in the morning after the surgery showed no acute nerve symptoms, and their ventricular size was compared with their preoperative one. Depending on the patient’s condition, a CT scan was performed one day to one month after the surgery to compare the postoperative with the preoperative ventricular size.
Some of the patients also serially underwent a CT scan. The bicaudate ratio was used to determine the hydrocephalus level. In case of decreases in the level of consciousness or acute nerve symptoms, brain CT scans were immediately performed to obtain the pathologic outcomes of the patients. After undergoing the surgery, the patients were asked to submit the results of their preoperative MRI and postoperative CT scan to be included in the study. The data were analysed in SPSS-25 using the Chi-square test, the t-test and the Mann-Whitney U test.
3. Results
The present study was conducted on 102 patients, including 64 (62.7%) males and 38 (37.3%) females, with a mean age of 28.9±2.23 years. The tumor lay in the third ventricle in 47.1% (n=48) of the patients, the fourth ventricle in 33.3% (n=34), the lateral ventricle in 13.7% (n=14), the third and fourth ventricles in 2% (n=2) and lateral and third ventricles in 3.9% (n=4). The obtained pathologic results suggested that colloid cysts in 28 cases, moduloblastoma in 14 and grade 2 ependymoma in 11 were the most prevalent types of the tumor. Comparing the results of the tumor pathology by age and gender according to figure 1 showed that GBM, meningium and SEGA were more prevalent in the women than in the men, choroid plexus papilloma was as prevalent in the men as in the women and the prevalence of the remaining cases was higher in the men. Significant tumor-related pathological abnormalities were also observed in both genders (P=0.26). Moreover, the tumor pathology was significant in terms of age in a way that ATRT and meningioma were the most prevalent in the youngest and oldest of the patients, respectively (P<0.001) (Figure 2).
Evaluating the tumor pathology and location also found colloidal cyst with a prevalence of 28 (100%) to be the most prevalent tumor in the third ventricle, medulloblastoma with a prevalence of 11 (78.6%) to be the most prevalent tumor in the fourth ventricle and astrocytoma with a prevalence of 3 (100%) to be the most prevalent in the lateral ventricles. The tumor pathology was significantly different in terms of its location (P<0.001) (figure 3).
Comparing the results of the 102 patients showed preoperative hydrocephalus in 81 (79.4%) patients, 41 (40.2%) of whom received a shunt. Hydrocephalus was observed in 35.3% (n=36) of the patients before and after the surgery, whereas it was not diagnosed in 15.7% (n=16) before the operation. Only postoperative, rather than preoperative, hydrocephalus was diagnosed in 4.9% (n=5) of the patients, whereas preoperative, rather than postoperative, hydrocephalus was diagnosed in 44.1% (n=45) of the patients.
Table 1 presents the frequency distribution of hydrocephalus before and after the surgery by age, gender, tumor location and tumor pathology. According to table 1 and the t-test results, despite the higher mean age of the patients with hydrocephalus, no significant differences were observed between the patients with and without hydrocephalus either before or after the surgery (P=0.78).
The prevalence of preoperative hydrocephalus was 81.3% in the men and 76.3% in the women (P=0.55), and that of postoperative hydrocephalus was 35.9% in the men and 47.4% in the women (P=0.26), suggesting no significant differences between the two genders. In terms of the tumor location, the prevalence of preoperative hydrocephalus was higher in the third and fourth ventricles and the third and lateral ventricles, although the difference was insignificant (P=0.32). In terms of the operative site, the third and fourth ventricles were the most prevalent sites of the tumor, although post-operative hydrocephalus was not significantly and statistically associated with the tumor location (P=0.17).
Preoperative hydrocephalus was pathologically observed in all the cases of arachnoid cysts, ATRT, GBM, meningium, grade 4 ependymoma and teratoma (P=0.2). Postoperative hydrocephalus was also reported in all the cases of GBM, grade 4 ependymoma and teratoma (P=0.2). The tumor pathology, however, did not show any significant differences in the incidence of hydrocephalus between before and after the surgery.
An EVD was used to improve hydrocephalus in 19 (18.6%) cases before the surgery and 71 (69.6%) afterwards. A total of 23.5% of the patients with hydrocephalus received an EVD, and significant differences were observed in the use of EVD before the operation (P=0.014). The frequency of EVD in operation was also 80.2% in the patients with hydrocephalus and 28.6% in those without hydrocephalus. Moreover, statistically-significant relationships were found between the use of postoperative EVD and the absence of preoperative hydrocephalus (P<0.001). Out of the 102 patients undergoing the surgery, 41 (40.2%) were treated with a shunt. The frequency of shunt insertion was 44.4% in the patients with hydrocephalus and 23.8% in those without hydrocephalus, suggesting no significant differences in shunt insertion between the two groups (P>0.066). Table 2 presents the frequency distribution of EVD use and shunt insertion in the patients with and without hydrocephalus.
According to table 3, the frequency of EVD was 20.8% in third ventricular tumors, 17.6% in fourth ventricular tumors, 100% in third and fourth ventricular tumors and 25% in third and lateral ventricular tumors, and the preoperative use of EVD was significantly different by the tumor location (0.016). The frequency of EVD usage was also significantly different by the tumor location, with 54.2% in third ventricular tumors, 85.3% in fourth ventricular tumors, 71.4% in lateral ventricular tumors and 100% in third ventricular tumors plus fourth ventricle and third+lateral ventricles (P=0.017). No significant differences were also observed in shunt insertion by the tumor location (P=0.17).
According to table 4 and the tumor pathology, no significant differences were observed in the distribution of EVD between before and after the operation.
4. Discussion
As a serious complication in patients with intraventricular tumors, hydrocephalus can exacerbate the disease symptoms, and cause death if not treated in a timely manner. Preventing hydrocephalus in patients undergoing a cerebral surgery is therefore considered a priority in postoperative care. Despite the importance of this complication, no comprehensive studies have yet been conducted in Iran on the incidence of hydrocephalus and the demographic and clinical features of the patients. The present study was conducted to determine demographic and clinical features and type of hydrocephalus therapeutic interventions before and after the surgery in patients with intraventricular brain tumors.
The present findings suggested that colloid cyst, medulloblastoma and grade 2 ependymoma were the most prevalent tumor in the patients with intraventricular tumors. In 2018, Pereira et al. also found colloid cysts to be the most prevalent intraventricular tumors [7]. The incidence and prevalence of these tumors have significantly increased in recent years due to different causes, including the increased risk factors for brain tumors, increased exposure to risk factors, advances in diagnostic and therapeutic facilities and new classifications of brain tumors [8].
Investigating demographic characteristics found below 10 years with a frequency of 31.4% to be the most prevalent age in the patients with intraventricular tumors. These tumors were also observed in over 60% of the males. Moreover, June et al. found the prevalence of intraventricular tumors to be higher in younger ages, the mean age of the patients to be 36 years and over two thirds of them to be male [9]. According to the results of the present study, the tumor pathology was not significantly different between the men and women, although it was significantly different among the different age groups. Contro et al. found the tumor pathology to be significantly different among the age groups of the patients, and the patients with SEGA and ATRT to be the youngest [10], which is consistent with the present study.
Investigating the tumor location and its associations with demographic characteristics found the third ventricle with a frequency of 47.1% to be the most prevalent site of intraventricular tumors. Furthermore, the tumor pathology was significantly related to the tumor location in a way that all the colloidal cysts lay in the third ventricle and 78.6% of the medulloblastomas lay in the fourth ventricle. Waldron et al. reported significant associations between the type and location of the tumor [8]. They found the prevalence of hydrocephalus to be 79.4% in the patients undergoing intraventricular tumor surgeries.
A hydrocephalic estimation in 2016 by Peyellow et al. showed that shunt insertion was more prevalent in patients younger than two years old, moderate-to-severe preoperative hydrocephalus, brain metastases and pathology of medulloblastoma and ependymoma compared to that in other cases [11]. In addition, a study by Guive et al. in 2014 on the endoscopic treatment of 53 patients with third-ventricle non-hydrocephalic colloid cysts found only two patients to require treatments for post-operative hydrocephalus [12]. A study conducted by Jay Talin et al. in 2015 on evaluating and treating hydrocephalus in children with posterior fossa tumors reported young age, preoperative hydrocephalus, metastasis, middle-line tumors, tumor pathology, subtotal tumor resection and EVD use in continuous post-operative hydrocephalic outbreaks [13].
A study conducted by Symss et al. in India in 2011 on 78 patients with colloid cysts found the prevalence of preoperative hydrocephalus to be 83.4%, which is consistent with the present research, although they found shunt insertion to be eventually required in four of the patients, which is less than the corresponding figure in the present study [14]. A study conducted by Cotton et al. in 2002 on 105 patients with colloid cysts reported shunt insertion requirements in 23 of the patients, which is consistent with the shunt insertion requirements in the present study [15]. The difference in the incidence of shunt insertion can be explained by the differences in the surgical technique used, the amount of surgical manipulation and intraoperative bleeding and the ability to completely remove colloid cysts without tearing the content of the cyst into the ventricle.
Using interhemispheric approaches and younger ages were found correlated with postoperative hydrocephalus [8], although it was not confirmed in the majority of similar studies. Furthermore, Philippe Kapley et al. reported different degrees of hydrocephalus in 29 patients with intraventricular tumors. They found that ventriculocystotomy alone was associated low success rates, and that it can eliminate the need for a shunt in the following 5 years in 83% of the cases if it is used in combination with endoscopic cisternotomy [9].
The present study found all the intra-cerebroventricular arachnoid cases to be hydrocephalus prior to the surgery, and half of the patients to need shunt insertion after the surgery. It is worth noting the open type of the surgical technique used and only performing the resection of the walls of the cysts. A study conducted by Eitan et al. in 2016 found 18% of patients diagnosed with intraventricular ependymoma in the fourth ventricle to need shunt insertion after the tumor resection [10], whereas the present study found this figure to be 27.3% in grade 2 ependymoma and 100% in grade 4.
5. Conclusion
Some features, including the pathology and location of the tumor, appeared effective in the subsequent hydrocephalic survival both before and after the operation. If these findings are confirmed and given these factors, therapeutic measures such as EVD and shunt insertion can be considered for preventing and treating postoperative hydrocephalus. Given the limitations of the present study, including a small sample, further research is recommended that be conducted using larger samples.
Ethical Considerations
Compliance with ethical guidelines
The present study was approved by the Ethics Committee of Isfahan University of Medical Sciences. Informed consent was obtained from all the participants.
Funding
This study was supported by Isfahan University of Medical Sciences. The study design was registered at the research faculty of the School of Medicine, Isfahan University of Medical Sciences (Code: 395112) The present article was partly extracted from a thesis of Mohammad Bagher Sadeghi on Neurosurgery.
Authors contributions
Study idea, design, interpretation and writing the paper: Saeid Abrishamkar; Data analysis and interpretation, preparing and writing the paper, submitting the manuscript and corresponding with the reviewers: Mohammad Bagher Sadeghi.
Conflict of interest
The authors declared no conflicts of interest regarding the publication of the present article.
Acknowledgements
The authors would like to express their gratitude to the authorities of School of Medicine, Isfahan University of Medical Sciences, for their efforts.
References
Jemal A, Siegel R, Xu J, Ward E. Cancer statistics, 2010. CA: A Cancer Journal for Clinicians. 2010; 60(5):277-300. [DOI:10.3322/caac.20073] [PMID]
Ohgaki H, Kleihues P. Population-based studies on incidence, survival rates, and genetic alterations in astrocytic and oligodendroglial gliomas. Journal of Neuropathology & Experimental Neurology. 2005; 64(6):479-89. [DOI:10.1093/jnen/64.6.479] [PMID]
Ge L, Hoa NT, Cornforth AN, Bota DA, Mai A, Kim DI, et al. Glioma big potassium channel expression in human cancers and possible T cell epitopes for their immuneotherapy. The Journal of Immunology. 2012; 189(5):2625-34. [DOI:10.4049/jimmunol.1102965] [PMID] [PMCID]
Burger PC, Schelthauer BW, Vogel FS,editors. Surgical pathology of the nervous system and its covering. 3rd ed. New York: Churchill Livingstone; 1991.
Brown PD, Decker PA, Rummans TA, Clark MM, Frost MH, Ballman KV, et al. A prospective study of quality of life in adults with newly diagnosed high-grade gliomas: Comparison of patient and caregiver ratings of quality of life. American Journal of Clinical Oncology. 2008; 31(2):163-8. [DOI:10.1097/COC.0b013e318149f1d3] [PMID]
Aaronson NK. Quality of life: What is it? How should it be measured? Oncol (Williston Park). 1988; 2(5):69-76.
Pereira BJ, de Almeida AN, Paiva WS, de Aguiar PH, Teixeira MJ, Marie SK. Natural history of intraventricular meningiomas: systematic review. Neurosurgical review. 2018; 15:1-11. [DOI:10.1007/s10143-018-1019-0]
Waldron JS, Tihan T. Epidemiology and pathology of intraventricular tumors. Neurosurgery Clinics. 2003; 14(4):469-82. [DOI:10.1016/S1042-3680(03)00060-3]
Brandsma D, van den Bent MJ. Molecular targeted therapies and chemotherapy in malignant gliomas. Current Opinion in Oncology. 2007; 19(6):598-605. [DOI:10.1097/CCO.0b013e3282f0313b] [PMID]
Jie NJ, Teo KA, Shabbir A, Yeo TT. Widespread intra-abdominal carcinomatosis from a rhabdoid meningioma after placement of a ventriculoperitoneal shunt: A case report and review of the literature. Asian journal of neurosurgery. 2018; 13(1):176-83. [DOI:10.4103/1793-5482.181128] [PMID] [PMCID]
Contro E, De Musso F, Pilu G, Ghi T. Intracranial Hemorrhage, Cysts, Tumors, and Destructive Lesions. In: Obstetric Imaging: Fetal Diagnosis and Care (2nd Edition) 2018 : 204-212. [DOI:10.1016/B978-0-323-44548-1.00040-1] [PMID] [PMCID]
Paul Foreman, m.D..Samuel mcclugage III, m.D., Robert naFtel, m.D. et al. Validation and modifcation of a predictive model of postresection hydrocephalus in pediatric patients with posterior fossa tumors. Journal of Neurosurgery: Pediatrics. 2013. 12(3):220-6. [DOI:10.3171/2013.5.PEDS1371] [PMID]
Guive Sharifi, Mehrdad Hosseinzadeh Bakhtevari, Omidvar Rezaei, Reza Jabbari: Endoscopic Surgery for Third Ventricular Colloid Cysts in the Absence of Hydrocephalus: a feasibility study. International Clinical Neuroscience Journal. 2014; 1(2):73-7.
Chih-Ta Lin & Jay K. Riva-Cambrin: Management of posterior fossa tumors and hydrocephalus in children: A review. Child's Nervous System. 2015; 31(10):1781-9. [DOI:10.1007/s00381-015-2781-8] [PMID]
Chih-Ta Lin & Jay K. Riva-Cambrin: Management of posterior fossa tumors and hydrocephalus in children: A review. Child's Nervous System. 2015; 31(10):1781-9. [DOI:10.1007/s00381-015-2781-8] [PMID]
Type of Study: Research |
Subject:
Brain Tumors
Send email to the article author
| Rights and Permissions | |
 |
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License. |