Sat, Jul 12, 2025
Volume 6, Issue 2 (Spring 2020)
Iran J Neurosurg 2020, 6(2): 57-66 |
Back to browse issues page
Download citation:
BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:



BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:
Aggarwal V, Jindal N, Rohilla S, . H, Singh I. Cerebrospinal Fluid Dynamics Study: A Unique Tool for Management of Chiari 1 Malformation Patients. Iran J Neurosurg 2020; 6 (2) :57-66
URL: http://irjns.org/article-1-212-en.html
URL: http://irjns.org/article-1-212-en.html
1- MS., MCh., Assistant Professor, Department of Neurosurgery, Pandit Bhagwat Dayal Sharma Post Graduate Institute of Medical Sciences, Rohtak, India
2- MD., Professor of Radiodiagnosis, Department of Radiodiagnosis, Pandit Bhagwat Dayal Sharma Post Graduate Institute of Medical Sciences, Rohtak, India
3- MS., MCh., Assistant Professor, Department of Neurosurgery, Pandit Bhagwat Dayal Sharma Post Graduate Institute of Medical Sciences, Rohtak, India ,drishwarsingh@yahoo.co.in
2- MD., Professor of Radiodiagnosis, Department of Radiodiagnosis, Pandit Bhagwat Dayal Sharma Post Graduate Institute of Medical Sciences, Rohtak, India
3- MS., MCh., Assistant Professor, Department of Neurosurgery, Pandit Bhagwat Dayal Sharma Post Graduate Institute of Medical Sciences, Rohtak, India ,
Full Text [PDF 2548 kb]
(1344 Downloads)
| Abstract (HTML) (3839 Views)
The clinical condition of the operated patients was followed up for 1 year.
MRI technique
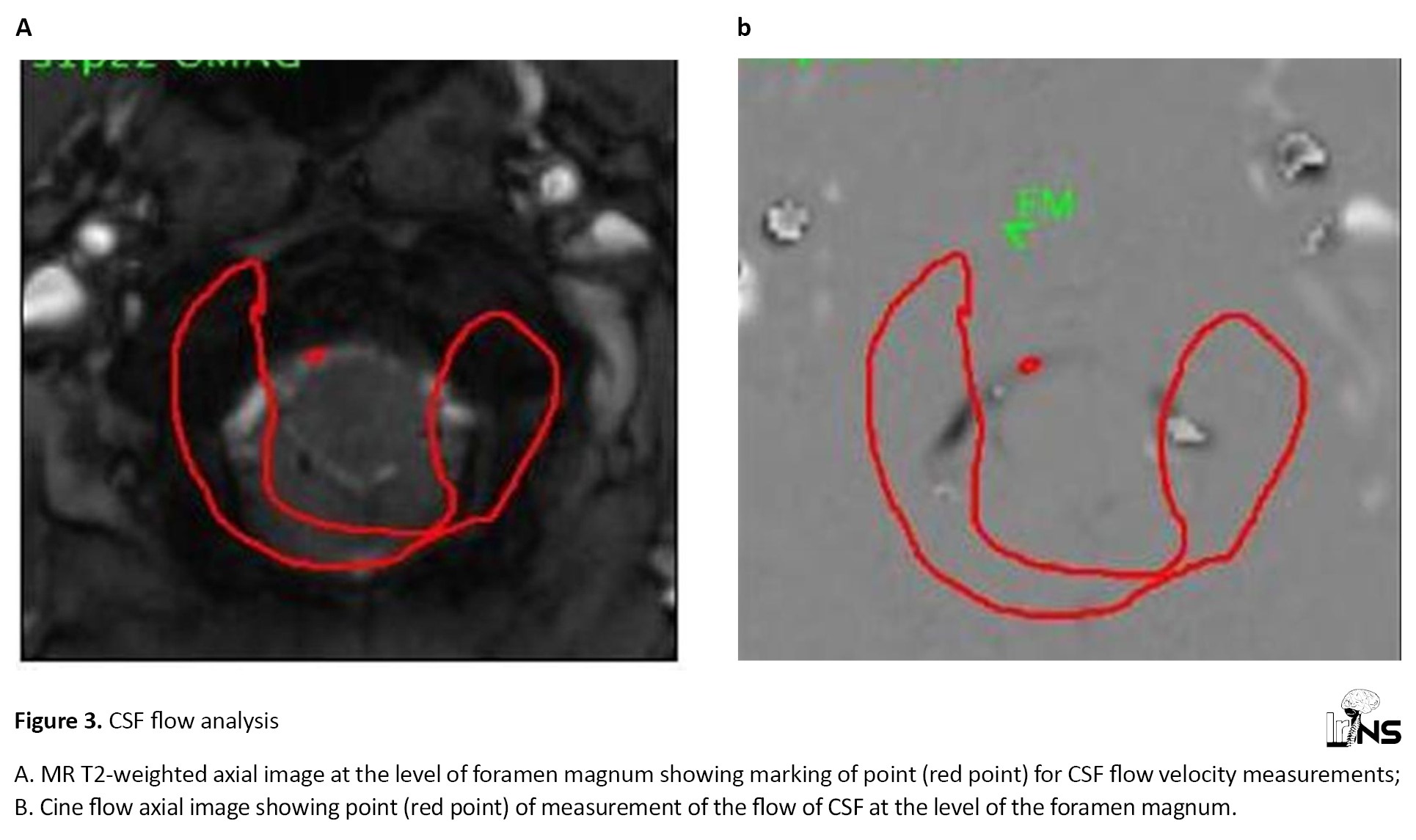
MRI was performed using a 3.0 Tesla MRI scanner (Discovery MR 750 w; GE, Ohio, USA). The imaging protocol included the sagittal and axial T2-weighted (W) scan for the morphological assessment and a low-velocity encoding (5 cm/sec) cine phase-contrast scans for imaging the CSF flow at the level of the foramen magnum. The scan parameters were as follows:
T2-weighted imaging-Slice thickness: 5mm, Slice spacing-: 1 mm, Bandwidth-: 50 kHz, TE-: 114 ms, TR-: 8901 ms, NEX-: 2.
CSF flow study-Slice thickness: 8 mm, Slice spacing-: 0, TR-: 26, Flip angle-: 10, Matrix-: 416×256, Bandwidth-: 13.89 kHz, NEX-:1.
Flow analysis was conducted using in-built software in the machine only (Figure 3).
Peak systolic and diastolic CSF flow velocities, as well as the flow rate of CSF, were measured pre-operatively at the cervico-medullary junction and post-operatively at 3 months in the same site.
Surgical technique
A total of 24 patients underwent surgical decompression of the foramen magnum with duraplasty. In the prone position, a standard midline suboccipital craniectomy with the C1 laminectomy was performed for wide decompression of cerebellar tonsils, brainstem, and spinal cord. The posterior atlanto-occipital membrane was cut and standard Y-shaped durotomy was done. The arachnoid membrane was opened, adhesions released and CSF flow was ensured. Then, the duraplasty was done using the pericranial patch or fascialata ensuring watertight closure. The wound was then closed in a standard multilayered fashion ensuring obliteration of any free space. One patient had obstructive hydrocephalus so only ventriculoperitoneal (VP) shunt was placed. One patient had progressive syringomyelia after decompression surgery, so a syringo-subarachnoid shunt was put afterward.
Statistical analysis
All numerical values were stored and analyzed by student t-test (paired and unpaired) using SPSS software version 26, 2019 (Statistical Package for the Social Sciences).
3. Results
The present study included 13 male and 12 female patients with an age ranged 13-60 years. The mean age at presentation was 33.5 years. All of the patients were symptomatic for CIM with wide variation in their clinical symptoms. Most of the patients (96%) had occipital headache as one of the presenting symptoms (Table 1).

Other symptoms were vertigo/dizziness (56%), pain in the neck (48%), and dysphagia (4%). The mean duration of symptoms was 2 years. The degree of tonsillar descent in the pre-operative period ranged from 6 to 32 mm. Syrinx was present in 11 patients (44%) and the most common location was the cervico-thoracic region (Table 2).

Out of the 11 patients with syrinx, 4 patients had associated scoliosis. The mean duration of the follow-up period was 1.3 years.
All of the patients had diminished CSF spaces anteriorly and posteriorly (cisterna magna) in the region of the foramen magnum. In the cine flow MRI, all of the patients had abnormal CSF flow at the foramen magnum region.
Clinical outcome
Occipital headache was relieved completely or partially in all 24 patients who underwent decompression surgery. Sixteen out of 24 patients (66.7%) had complete resolution of headache and 8 patients (33.3%) had a partial resolution. One patient with VP shunt surgery had complete resolution of the headache. Other symptoms like vertigo and dysphagia were completely relieved in all of the respective patients. Dysaesthetic pain alleviated in all patients with syringomyelia except one patient who had progressive syringomyelia even after decompression surgery. This patient with progressive syringomyelia developed mild quadriparesis after one month of decompression surgery and a syringo-subarachnoid shunt was placed. All other patients did not report any new symptoms or signs in the follow-up period post-operatively.
Radiological outcome
Tonsillar herniation was reduced and tonsils became round-shaped in all 24 patients of decompression surgery. One patient, who had VP shunt surgery, had persistent tonsillar herniation. Ten out of 11 patients with syrinx had a significant reduction in syrinx diameter. Only 1 patient showed an increase in syrinx diameter in the post-operative period. In cine flow MRI, the mean Peak Systolic Velocity (PSV) of CSF flow at foramen magnum was 3.60080 cm/sec in the pre-operative period and reduced to 1.89320 cm/sec in the post-operative period and the difference was statistically significant (P<0.05) as shown in Table 3.
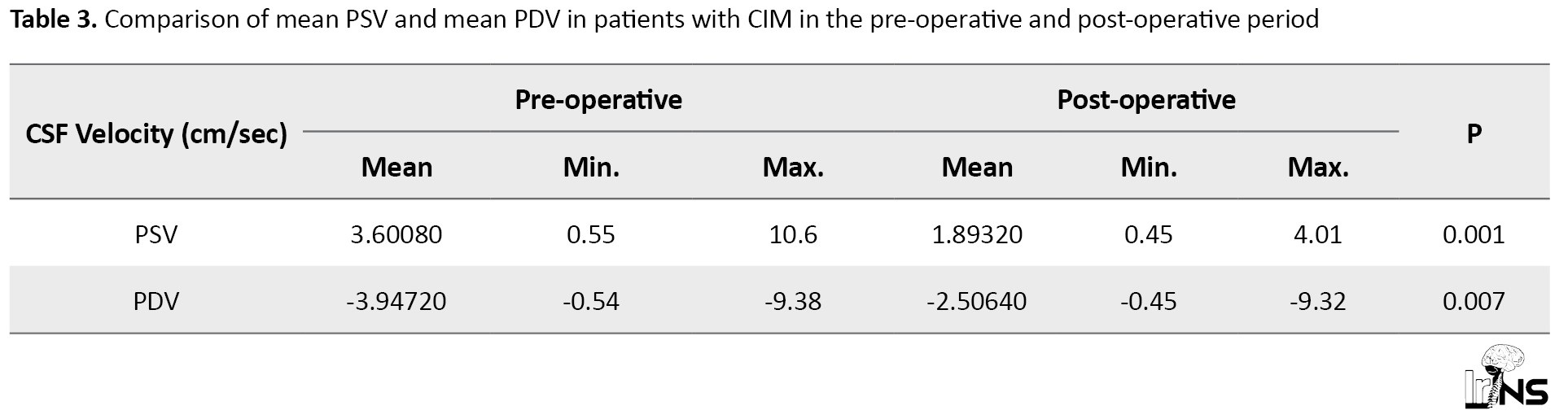
Similarly, the mean peak diastolic velocity (PDV) of CSF flow reduced from -3.94720 cm/s to -2.50640 cm/sec in the post-operative period, and the difference was found statistically significant (P<0.05).
4. Discussion
The initial documented case reports of Chiari malformation were narrated by Hans Chiari in 1891 [9]. Traditionally, they were classified into 4 types (I to IV), with types I and II constituting almost 99% of the cases [10]. Recently, Chiari 0 malformation and Chiari 1.5 malformation have been added to the list [11]. Chiari I malformation consists of 5 mm or more caudal descent of the cerebellar tonsils below the foramen magnum with or without associated syringomyelia and hydrocephalus (10%). Many hypotheses have tried to clarify the pathogenesis of the CIM [12, 13]. Tonsillar herniation which is the main component of CIM can be congenital or acquired.
The congenital nature of CIM has been explained based on craniocerebral disproportion i.e. small posterior fossa compared with the tissue volume [6, 12]. Presently, one-third of cases are detected incidentally due to the widespread use of MRI [5, 6]. Many of these cases are asymptomatic and in fact, it is proven that the degree of tonsillar descent has no relation with the clinical presentation in CIM [5, 6, 14]. Abnormal CSF flow dynamics in the region of the foramen magnum, due to the crowded posterior fossa, is the main pathophysiological factor responsible for progressive tonsillar descent, syrinx formation, and hence clinical symptoms of the patients [5, 6].
It is a well-known fact that there is a bidirectional cranio-spinal flow of CSF during each cardiac cycle [5, 6] (Figure 4).
The increased cerebral blood volume during systole displaces the CSF caudally through the foramen magnum and the elastic recoil of the spinal dura during diastole pushes the CSF back toward the cranial compartment [5]. In CIM with each systole, an abnormal high-velocity CSF jet across the foramen magnum propels the tonsils more caudally and this piston-like movement of tonsils is responsible for syrinx formation [15]. Cine flow MRI is one of the non-invasive means of studying CSF flow dynamics in the region of the foramen magnum. In the present study, we also found high mean PSV on cine flow MRI in the pre-operative period which reduced significantly in the post-operative period with clinico-radiological improvement, and the difference was found statistically significant. But the mean peak velocity values could not be compared with the previous studies because of the different methods employed for measuring these values on cine flow MRI [16, 17]. As all our patients had the CSF flow study done by the same senior radiologist so limiting factor of inter-observer variability seen in previous studies was eliminated in our study [6].
Literature also supports the presence of a direct correlation between altered CSF dynamics at foramen magnum with the clinical symptoms in CIM patients [7, 18]. Ventureyra et al. (2003), performed a study on the role of CSF flow dynamics using cine flow MRI in 24 patients with CIM [5]. They found a direct correlation between abnormal CSF dynamics and clinical symptoms of the patients and all 16 symptomatic patients relieved after the re-establishment of proper CSF flow at the foramen magnum. In our study, also 24 patients underwent decompression surgery for re-establishment of normal CSF flow in the region of the foramen magnum, and the symptoms were relieved in 23 out of 24 patients, though 33% of the patients had a mild headache in the follow-up period which could be due to craniectomy. Reports of diminished syrinx cavity have also been documented in previous studies after the restoration of proper CSF circulation.
Kumar et al. (2019), reported syrinx in 23 (71.87%) patients out of 32 patients of CIM [6]. After decompression surgery, 87% of patients with syrinx (20/23) demonstrated radiographic improvement and 13% remained stable or developed an increase in syrinx size. In our study, 11 out of 25 patients (44%) had syrinx and in post-operative MRI, 10 patients (91%) had a significant reduction in syrinx cavity diameter. One patient had progressive syringomyelia. Thus, the improvement in CSF flow dynamics correlated well with clinical and radiological improvement in patients with CIM.
5. Conclusion
Although abnormal CSF circulation is the most important pathognomonic factor responsible for the progression of CIM, still a large prospective follow-up study on asymptomatic patients is needed to correlate abnormal CSF flow dynamics with the occurrence of clinical symptoms and radiological changes. With these encouraging findings and review of literature, in future cine flow MRI may serve as an investigation of choice for proper selection of surgical candidates among asymptomatic patients with CIM. Improved CSF flow in cine flow MRI can predict good clinical and radiological outcomes.
Abnormal CSF flow dynamics are responsible for the progression of the disease and symptoms in CIM patients. The degree of tonsillar descent has no significant relationship with the occurrence of clinical symptoms in these patients. Cine flow MRI is a very valuable tool for the management of symptomatic patients with CIM.
Ethical Considerations
Compliance with ethical guidelines
The institutional review board of Postgraduate Institute of Medical Sciences, Rohtak has confirmed the study (Ethical Code: NS/18/655).
Funding
This article was extracted from the thesis with the title “To Study the CSF Flow in Patients of Chiari Malformation Type I using Cine MRI” which was defended in Pandit Bhagwat Dayal Sharma Post Graduate Institute of Medical Sciences, Rohtak, India.
Authors' contributions
Conceptualization and methodology: Navodhya Jindal, Seema Rohilla, Ishwar Singh; Data collection and data analysis: Varun Aggarwal, Navodhya Jindal, Seema Rohilla, Hitesh; Writing – original draft: Varun Aggarwal, Ishwar Singh; Critically revising the article: Varun Aggarwal, Ishwar Singh; Writing – review & editing: Ishwar Singh; Approving the final version of the manuscript: All authors
Conflict of interest
The authors declared no conflict of interest.
References
Full Text: (2025 Views)
1. Introduction
onsillar herniation of 5 mm or more below the foramen magnum, with or without associated syrinx, is called Chiari I Malformation (CIM) [1, 2]. Due to the extensive use of Magnetic Resonance Imaging (MRI), an increasing number of cases of tonsillar herniation is now detected incidentally. Approximately, one-third of individuals with tonsillar herniation revealed by MRI are asymptomatic [3, 4]. The degree of tonsillar herniation alone correlates poorly with the progression of CIM, clinical presentation, and surgical outcome [5, 6]. Relatively small posterior fossa leads to altered Cerebrospinal Fluid (CSF) dynamics in the region of the foramen magnum with each cardiac cycle.
The altered CSF dynamics is the main pathophysiological factor responsible for the progression of tonsillar descent, syrinx formation, and hence clinical presentation. That is why CSF dynamics study is nowadays the focus of research in not only symptomatic patients but also in asymptomatic patients with CIM.
Phase-contrast cine flow MRI is one of the non-invasive tools for studying CSF dynamics in the brain and at the foramen magnum. Although a large number of studies have proven the role of abnormal CSF circulation in symptomatic patients with CIM, they have their shortcomings like small sample size, short follow-up, inter-observer variability, etc. [5, 6, 7, 8]. After a comprehensive review of the literature and understanding the shortcomings of the previous studies, we performed this study to investigate the correlation between CSF dynamics at the foramen magnum and clinicoradiological profile of patients before and after foramen magnum decompression. The main aim of this study is to emphasize the role of abnormal CSF circulation in CIM for young neurosurgeons.
2. Methods & Materials/Patients
This prospective cohort study was carried out in the department of neurosurgery in collaboration with the department of radio-diagnosis and imaging between January 2018 and August 2019 after obtaining approval from the institutional review board of Postgraduate Institute of Medical Sciences, Rohtak, India. Written informed consent was obtained from all the patients. Only patients who met the criteria for CIM (peg-shaped cerebellar tonsils herniating 5 mm or more below the foramen magnum) without any intracranial space-occupying lesion were included in the study. The clinical symptoms and signs of all 25 patients were noted both pre-operatively and post-operatively. Plain MRI of brain and spine with cine flow MRI at Cranio-Vertebral Junction (CVJ) of all the patients was performed both pre-operatively and post-operatively at 3 months. Changes in CSF flow dynamic parameters and clinicoradiological improvement after surgery were noted and correlated (Figures 1 and 2).
onsillar herniation of 5 mm or more below the foramen magnum, with or without associated syrinx, is called Chiari I Malformation (CIM) [1, 2]. Due to the extensive use of Magnetic Resonance Imaging (MRI), an increasing number of cases of tonsillar herniation is now detected incidentally. Approximately, one-third of individuals with tonsillar herniation revealed by MRI are asymptomatic [3, 4]. The degree of tonsillar herniation alone correlates poorly with the progression of CIM, clinical presentation, and surgical outcome [5, 6]. Relatively small posterior fossa leads to altered Cerebrospinal Fluid (CSF) dynamics in the region of the foramen magnum with each cardiac cycle.
The altered CSF dynamics is the main pathophysiological factor responsible for the progression of tonsillar descent, syrinx formation, and hence clinical presentation. That is why CSF dynamics study is nowadays the focus of research in not only symptomatic patients but also in asymptomatic patients with CIM.
Phase-contrast cine flow MRI is one of the non-invasive tools for studying CSF dynamics in the brain and at the foramen magnum. Although a large number of studies have proven the role of abnormal CSF circulation in symptomatic patients with CIM, they have their shortcomings like small sample size, short follow-up, inter-observer variability, etc. [5, 6, 7, 8]. After a comprehensive review of the literature and understanding the shortcomings of the previous studies, we performed this study to investigate the correlation between CSF dynamics at the foramen magnum and clinicoradiological profile of patients before and after foramen magnum decompression. The main aim of this study is to emphasize the role of abnormal CSF circulation in CIM for young neurosurgeons.
2. Methods & Materials/Patients
This prospective cohort study was carried out in the department of neurosurgery in collaboration with the department of radio-diagnosis and imaging between January 2018 and August 2019 after obtaining approval from the institutional review board of Postgraduate Institute of Medical Sciences, Rohtak, India. Written informed consent was obtained from all the patients. Only patients who met the criteria for CIM (peg-shaped cerebellar tonsils herniating 5 mm or more below the foramen magnum) without any intracranial space-occupying lesion were included in the study. The clinical symptoms and signs of all 25 patients were noted both pre-operatively and post-operatively. Plain MRI of brain and spine with cine flow MRI at Cranio-Vertebral Junction (CVJ) of all the patients was performed both pre-operatively and post-operatively at 3 months. Changes in CSF flow dynamic parameters and clinicoradiological improvement after surgery were noted and correlated (Figures 1 and 2).
The clinical condition of the operated patients was followed up for 1 year.
MRI technique
MRI was performed using a 3.0 Tesla MRI scanner (Discovery MR 750 w; GE, Ohio, USA). The imaging protocol included the sagittal and axial T2-weighted (W) scan for the morphological assessment and a low-velocity encoding (5 cm/sec) cine phase-contrast scans for imaging the CSF flow at the level of the foramen magnum. The scan parameters were as follows:
T2-weighted imaging-Slice thickness: 5mm, Slice spacing-: 1 mm, Bandwidth-: 50 kHz, TE-: 114 ms, TR-: 8901 ms, NEX-: 2.
CSF flow study-Slice thickness: 8 mm, Slice spacing-: 0, TR-: 26, Flip angle-: 10, Matrix-: 416×256, Bandwidth-: 13.89 kHz, NEX-:1.
Flow analysis was conducted using in-built software in the machine only (Figure 3).
Peak systolic and diastolic CSF flow velocities, as well as the flow rate of CSF, were measured pre-operatively at the cervico-medullary junction and post-operatively at 3 months in the same site.
Surgical technique
A total of 24 patients underwent surgical decompression of the foramen magnum with duraplasty. In the prone position, a standard midline suboccipital craniectomy with the C1 laminectomy was performed for wide decompression of cerebellar tonsils, brainstem, and spinal cord. The posterior atlanto-occipital membrane was cut and standard Y-shaped durotomy was done. The arachnoid membrane was opened, adhesions released and CSF flow was ensured. Then, the duraplasty was done using the pericranial patch or fascialata ensuring watertight closure. The wound was then closed in a standard multilayered fashion ensuring obliteration of any free space. One patient had obstructive hydrocephalus so only ventriculoperitoneal (VP) shunt was placed. One patient had progressive syringomyelia after decompression surgery, so a syringo-subarachnoid shunt was put afterward.
Statistical analysis
All numerical values were stored and analyzed by student t-test (paired and unpaired) using SPSS software version 26, 2019 (Statistical Package for the Social Sciences).
3. Results
The present study included 13 male and 12 female patients with an age ranged 13-60 years. The mean age at presentation was 33.5 years. All of the patients were symptomatic for CIM with wide variation in their clinical symptoms. Most of the patients (96%) had occipital headache as one of the presenting symptoms (Table 1).

Other symptoms were vertigo/dizziness (56%), pain in the neck (48%), and dysphagia (4%). The mean duration of symptoms was 2 years. The degree of tonsillar descent in the pre-operative period ranged from 6 to 32 mm. Syrinx was present in 11 patients (44%) and the most common location was the cervico-thoracic region (Table 2).

Out of the 11 patients with syrinx, 4 patients had associated scoliosis. The mean duration of the follow-up period was 1.3 years.
All of the patients had diminished CSF spaces anteriorly and posteriorly (cisterna magna) in the region of the foramen magnum. In the cine flow MRI, all of the patients had abnormal CSF flow at the foramen magnum region.
Clinical outcome
Occipital headache was relieved completely or partially in all 24 patients who underwent decompression surgery. Sixteen out of 24 patients (66.7%) had complete resolution of headache and 8 patients (33.3%) had a partial resolution. One patient with VP shunt surgery had complete resolution of the headache. Other symptoms like vertigo and dysphagia were completely relieved in all of the respective patients. Dysaesthetic pain alleviated in all patients with syringomyelia except one patient who had progressive syringomyelia even after decompression surgery. This patient with progressive syringomyelia developed mild quadriparesis after one month of decompression surgery and a syringo-subarachnoid shunt was placed. All other patients did not report any new symptoms or signs in the follow-up period post-operatively.
Radiological outcome
Tonsillar herniation was reduced and tonsils became round-shaped in all 24 patients of decompression surgery. One patient, who had VP shunt surgery, had persistent tonsillar herniation. Ten out of 11 patients with syrinx had a significant reduction in syrinx diameter. Only 1 patient showed an increase in syrinx diameter in the post-operative period. In cine flow MRI, the mean Peak Systolic Velocity (PSV) of CSF flow at foramen magnum was 3.60080 cm/sec in the pre-operative period and reduced to 1.89320 cm/sec in the post-operative period and the difference was statistically significant (P<0.05) as shown in Table 3.

Similarly, the mean peak diastolic velocity (PDV) of CSF flow reduced from -3.94720 cm/s to -2.50640 cm/sec in the post-operative period, and the difference was found statistically significant (P<0.05).
4. Discussion
The initial documented case reports of Chiari malformation were narrated by Hans Chiari in 1891 [9]. Traditionally, they were classified into 4 types (I to IV), with types I and II constituting almost 99% of the cases [10]. Recently, Chiari 0 malformation and Chiari 1.5 malformation have been added to the list [11]. Chiari I malformation consists of 5 mm or more caudal descent of the cerebellar tonsils below the foramen magnum with or without associated syringomyelia and hydrocephalus (10%). Many hypotheses have tried to clarify the pathogenesis of the CIM [12, 13]. Tonsillar herniation which is the main component of CIM can be congenital or acquired.
The congenital nature of CIM has been explained based on craniocerebral disproportion i.e. small posterior fossa compared with the tissue volume [6, 12]. Presently, one-third of cases are detected incidentally due to the widespread use of MRI [5, 6]. Many of these cases are asymptomatic and in fact, it is proven that the degree of tonsillar descent has no relation with the clinical presentation in CIM [5, 6, 14]. Abnormal CSF flow dynamics in the region of the foramen magnum, due to the crowded posterior fossa, is the main pathophysiological factor responsible for progressive tonsillar descent, syrinx formation, and hence clinical symptoms of the patients [5, 6].
It is a well-known fact that there is a bidirectional cranio-spinal flow of CSF during each cardiac cycle [5, 6] (Figure 4).
The increased cerebral blood volume during systole displaces the CSF caudally through the foramen magnum and the elastic recoil of the spinal dura during diastole pushes the CSF back toward the cranial compartment [5]. In CIM with each systole, an abnormal high-velocity CSF jet across the foramen magnum propels the tonsils more caudally and this piston-like movement of tonsils is responsible for syrinx formation [15]. Cine flow MRI is one of the non-invasive means of studying CSF flow dynamics in the region of the foramen magnum. In the present study, we also found high mean PSV on cine flow MRI in the pre-operative period which reduced significantly in the post-operative period with clinico-radiological improvement, and the difference was found statistically significant. But the mean peak velocity values could not be compared with the previous studies because of the different methods employed for measuring these values on cine flow MRI [16, 17]. As all our patients had the CSF flow study done by the same senior radiologist so limiting factor of inter-observer variability seen in previous studies was eliminated in our study [6].
Literature also supports the presence of a direct correlation between altered CSF dynamics at foramen magnum with the clinical symptoms in CIM patients [7, 18]. Ventureyra et al. (2003), performed a study on the role of CSF flow dynamics using cine flow MRI in 24 patients with CIM [5]. They found a direct correlation between abnormal CSF dynamics and clinical symptoms of the patients and all 16 symptomatic patients relieved after the re-establishment of proper CSF flow at the foramen magnum. In our study, also 24 patients underwent decompression surgery for re-establishment of normal CSF flow in the region of the foramen magnum, and the symptoms were relieved in 23 out of 24 patients, though 33% of the patients had a mild headache in the follow-up period which could be due to craniectomy. Reports of diminished syrinx cavity have also been documented in previous studies after the restoration of proper CSF circulation.
Kumar et al. (2019), reported syrinx in 23 (71.87%) patients out of 32 patients of CIM [6]. After decompression surgery, 87% of patients with syrinx (20/23) demonstrated radiographic improvement and 13% remained stable or developed an increase in syrinx size. In our study, 11 out of 25 patients (44%) had syrinx and in post-operative MRI, 10 patients (91%) had a significant reduction in syrinx cavity diameter. One patient had progressive syringomyelia. Thus, the improvement in CSF flow dynamics correlated well with clinical and radiological improvement in patients with CIM.
5. Conclusion
Although abnormal CSF circulation is the most important pathognomonic factor responsible for the progression of CIM, still a large prospective follow-up study on asymptomatic patients is needed to correlate abnormal CSF flow dynamics with the occurrence of clinical symptoms and radiological changes. With these encouraging findings and review of literature, in future cine flow MRI may serve as an investigation of choice for proper selection of surgical candidates among asymptomatic patients with CIM. Improved CSF flow in cine flow MRI can predict good clinical and radiological outcomes.
Abnormal CSF flow dynamics are responsible for the progression of the disease and symptoms in CIM patients. The degree of tonsillar descent has no significant relationship with the occurrence of clinical symptoms in these patients. Cine flow MRI is a very valuable tool for the management of symptomatic patients with CIM.
Ethical Considerations
Compliance with ethical guidelines
The institutional review board of Postgraduate Institute of Medical Sciences, Rohtak has confirmed the study (Ethical Code: NS/18/655).
Funding
This article was extracted from the thesis with the title “To Study the CSF Flow in Patients of Chiari Malformation Type I using Cine MRI” which was defended in Pandit Bhagwat Dayal Sharma Post Graduate Institute of Medical Sciences, Rohtak, India.
Authors' contributions
Conceptualization and methodology: Navodhya Jindal, Seema Rohilla, Ishwar Singh; Data collection and data analysis: Varun Aggarwal, Navodhya Jindal, Seema Rohilla, Hitesh; Writing – original draft: Varun Aggarwal, Ishwar Singh; Critically revising the article: Varun Aggarwal, Ishwar Singh; Writing – review & editing: Ishwar Singh; Approving the final version of the manuscript: All authors
Conflict of interest
The authors declared no conflict of interest.
References
- Chauvet D, Carpentier A, George B. Dura splitting decompression in Chiari type 1 malformation: Clinical experience and radiological findings. Neurosurgical Review. 2009; 32:465-70. [DOI:10.1007/s10143-009-0214-4] [PMID]
- Ball WS, Crone KR. Chiari I malformation: From Dr. Chiari to MR imaging. Radiology. 1995; 195:602-4. [DOI:10.1148/radiology.195.3.7753980] [PMID]
- Meadows J, Kraut M, Guarnieri M, Haroun RI, Carson BS. Asymptomatic Chiari type I malformations identified on magnetic resonance imaging. Journal of Neurosurgery. 2000; 92:920-6. [DOI:10.3171/jns.2000.92.6.0920] [PMID]
- Elster AD, Chen MY. Chiari I malformation: Clinical and radiological appraisal. Radiology. 1992; 183:347-53. [DOI:10.1148/radiology.183.2.1561334] [PMID]
- Ventureyra EC, Aziz HA, Vassilyadi M. The role of cine flow MRI in children with Chiari I malformation. Child's Nervous System. 2003; 19:109-13. [DOI:10.1007/s00381-002-0701-1] [PMID]
- Kumar A, Ghosh SN, Sadique SI. Clinicoradiological study of adult Chiari malformation type 1 patients with emphasis on cerebrospinal fluid peak flow velocity at foramen magnum level. Neurology India. 2019; 67(3):744-8. [DOI:10.4103/0028-3886.263214] [PMID]
- Panigrahi M, Reddy BP, Reddy AK, Reddy JJ. CSF flow study in Chiari I malformation. Child's Nervous System. 2004; 20(5):336-40. [DOI:10.1007/s00381-003-0881-3] [PMID]
- Roldan A, Wieben O, Haughton V, Osswald T, Chesler N. Characterization of CSF hydrodynamics in the presence and absence of tonsillar ectopia by means of computational flow analysis. American Journal of Neuroradiology. 2009; 30(5):941-6. [DOI:10.3174/ajnr.A1489] [PMID] [PMCID]
- Chiari H. Uber Veranderungen des Kleinhirnsinfolge von Hydrocephalie des Grosshirns. DMW-Deutsche Medizinische Wochenschrift. 1891; 17(42):1172-5. [DOI:10.1055/s-0029-1206803]
- Ozsoy KM, Oktay K, Cetinalp NE, Gezercan Y, Erman T. The role of cine flow magnetic resonance imaging in patients with Chiari 0 malformation. Turk Neurosurg. 2018; 28(2):251-6. [DOI: 10.5137/1019-5149.JTN.19049-16.2]
- Tubbs RS, Oakes WJ. Chiari malformations. In: Winn HR, editor. Youmans & Winn neurological surgery, 7th ed. Philadelphia PA: Elsevier; 2017.
- Oldfield EH. Pathogenesis of Chiari I- Pathophysiology of Syringomyelia: Implications for therapy: A summary of 3 decades of clinical research. Neurosurgery. 2017; 64(1):66-77. [DOI:10.1093/neuros/nyx377] [PMID]
- Sekula Jr RF, Arnone GD, Crocker C, Aziz KM, Alperin N. The pathogenesis of Chiari I malformation and syringomyelia. Neurological Research. 2011; 33(3):232-9. [DOI:10.1179/016164111X12962202723724] [PMID]
- Milhorat TH, Chou MW, Trinidad EM, Kula RW. Chiari I malformation redefined: Clinical and radiographic findings for 364 symptomatic patients. Neurosurgery. 1999; 44(5):1005-17. [DOI:10.1097/00006123-199905000-00042] [PMID]
- Oldfield EH, Muraszko K, Shawker TH, Patronas NJ. Pathophysiology of syringomyelia associated with Chiari I malformation of the cerebellar tonsils. Implications for diagnosis and treatment. Journal of Neurosurgery 1994; 80(1):3-15. [DOI:10.3171/jns.1994.80.1.0003] [PMID]
- Armonda RA, Citrin CM, Foley KT, Ellenbogen RG. Quantitative cine-mode magnetic resonance imaging of Chiari I malformations: An analysis of cerebrospinal fluid dynamics. Neurosurgery. 1994; 35(2):214-24. [DOI:10.1227/00006123-199408000-00006] [PMID]
- Heiss SD, Patronas N, DeVroom HL, Shawker T, Ennis R, Kammerer W, et al. Elucidiating the pathophysiology of syringomyelia. Journal of Neurosurgery. 1999; 91(4):553-62. [DOI:10.3171/jns.1999.91.4.0553] [PMID]
- Greitz D, Wirestam R, Frank A, Nordell B, Thomsen C, Stahlberg F. Pulsatile brain movement and associated hydrodynamics studied by magnetic resonance phase imaging. Neuroradiology. 1992; 34(5):370-80. [DOI:10.1007/BF00596493] [PMID]
Type of Study: Research |
Subject:
Basic Neurosurgery
Send email to the article author
| Rights and Permissions | |
 |
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License. |






.jpg)
.jpg)