Tue, Jul 15, 2025
Volume 3, Issue 3 (12-2017)
Iran J Neurosurg 2017, 3(3): 103-108 |
Back to browse issues page
Download citation:
BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:



BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:
Yousefzadeh-Chabok S, Kazemnejad-Leili E, Kouchakinejad-Eramsadati L, Moghtader M, Abolfathi N, Reihanian Z et al . Hydrocephalus in Patients With Head Trauma: A Series of 14 Patients. Iran J Neurosurg 2017; 3 (3) :103-108
URL: http://irjns.org/article-1-113-en.html
URL: http://irjns.org/article-1-113-en.html
Shahrokh Yousefzadeh-Chabok1 
 , Ehsan Kazemnejad-Leili2
, Ehsan Kazemnejad-Leili2 
 , Leila Kouchakinejad-Eramsadati
, Leila Kouchakinejad-Eramsadati 
 , Maria Moghtader3
, Maria Moghtader3 
 , Nasim Abolfathi3
, Nasim Abolfathi3 
 , Zoheir Reihanian4
, Zoheir Reihanian4 
 , Marieh Hosseinpour *
, Marieh Hosseinpour * 



 , Ehsan Kazemnejad-Leili2
, Ehsan Kazemnejad-Leili2 
 , Leila Kouchakinejad-Eramsadati
, Leila Kouchakinejad-Eramsadati 
 , Maria Moghtader3
, Maria Moghtader3 
 , Nasim Abolfathi3
, Nasim Abolfathi3 
 , Zoheir Reihanian4
, Zoheir Reihanian4 
 , Marieh Hosseinpour *
, Marieh Hosseinpour * 


1- 1 Neuroscience Research Center, Guilan University of Medical Sciences, Rasht, Iran. 2 Guilan Road Trauma Research Center, Guilan University of Medical Sciences, Rasht, Iran.
2- Guilan Road Trauma Research Center, Guilan University of Medical Sciences, Rasht, Iran.
3- Department of Nursing, Poursina Hospital, Guilan University of Medical Sciences, Rasht, Iran
4- Department of Neurosurgery, Poursina Hospital, Guilan University of Medical Sciences, Rasht, Iran
2- Guilan Road Trauma Research Center, Guilan University of Medical Sciences, Rasht, Iran.
3- Department of Nursing, Poursina Hospital, Guilan University of Medical Sciences, Rasht, Iran
4- Department of Neurosurgery, Poursina Hospital, Guilan University of Medical Sciences, Rasht, Iran
Full Text [PDF 592 kb]
(2985 Downloads)
| Abstract (HTML) (6708 Views)
The outcome of patients was evaluated based on the GOS and categorized in five groups of complete recovery, mild disability, moderate disability, severe disability, vegetative state, and death. Accordingly, 21.4% (n=3) had complete recovery. Moderate disability was found in 21.4% (n=3), severe disability in 14.3% (n=2), vegetative state in 7.14% (n=1), and death in 35.7% (n=5). Independent T-test demonstrated that no statistically significant relationship was observed between age and hydrocephalus (P=0.06), but there was a statistically significant relationship between GCS and hydrocephalus (P=0.03). Also, a statistically significant relationship was found between GCS and hydrocephalus (P=0.063) and the mechanism of trauma and hydrocephalus (P=0.817) using Fisher’s exact test showed that no statistically significant relationship was demonstrated between these variables and hydrocephalus incidence. In addition, the results of the analysis of the test about the association between accompanying cranial damages and hydrocephalus found that only ICH (P=0.045) and IVH (P=0.013) on admission had a statistically significant relationship with hydrocephalus incidence.
4. Discussion
Ventricular dilatation is a common complication in patients with TBI that can be seen in subsequent examinations on the patient and can become PTH [18]. If not diagnosed and treated, it can negatively affect the patient’s recovery and outcome [2]. Based on the results of our study, the frequency of hydrocephalus in 548 patients with head injury was obtained 14 patients (2.6%). In a study by Choi et al., 28 out of 693 (4%) patients with trauma were found to have hydrocephalus [4]. The study results of Kim et al. examining 789 patients with head injury showed that hydrocephalus was detected in 129 patients [19]. In a study on 140 patients with severe TBI, Mazzini and colleagues found that 45% of 140 patients manifested degrees of hydrocephalus, but severe hydrocephalus was found in only 11% of patients [20].
In another study by Tian et al., 36 out of 301 patients (11.96%) suffered from hydrocephalus during a three month period [16]. The mean age of the patients in this study was 44.07±24.48 years old, ranging from 7 to 83 years old. The mean age of the patients was 33.7±16 years, with the range of 2 to 65 years, in the study by Sarkari et al. [13]. In another study, the age range of patients was 2 to 89 years, and the mean age was 49.7±10.5 years [19]. In another study, the mean age of patients was 36±16 with the age range of 15 to 79 years old [20]. Bhatoe and his colleague reported the age range of patients 12 to 65 years [21].
In this study, 31.1% of men (n=14) suffered from hydrocephalus, while women showed no case of hydrocephalus. Other studies demonstrated that men had more hydrocephalus cases compared to women [13, 21]. Crashes (85.7%) and fall (14.3%) were identified as the two leading traumatic causes of hydrocephalus in patients. In another study, road accidents (68%) and then height fall (32%) were reported as the most common causes of injury [13]. In this study, head injury severity was found to be mild in most patients with hydrocephalus (n=7) (GCS=13-15) and then moderate (n=6) (GCS=9-12), and severe (n=1) (GCS=3-8). In another study, most patients (68%) had severe head injuries on admission (GCS less than or equal to 8) [13].
In a study by Kim et al., GCS on admission was categorized in four groups; less than 7 in 39 patients, between 8 and 10 in 13 patients, between 11 to 13 in 9 patients, and between 14 and 15 in 3 patients. Hydrocephalus occurred significantly higher in patients with low GCS on admission [19]. Another study showed that the mean GCS on admission in hydrocephalic patients was 13±2.8, ranging from 6 to 15 [20]. In another study, 25 patients with hydrocephalus caused by post-traumatic injury were examined. It was found that 20 patients had closed head injury. In these patients, the primary head injury in 15 patients was severe (GCS≤8) [21]. In our study, the most frequent accompanying cranial injuries observed in CT scan images were SAH (n=5), then EDH (n=4), and ICH (n=4). In another study on patients with hydrocephalus, 28 patients (76%) suffered from brain contusion, 19(50%) SAH, 18(47%) SDH, 3(8%) IVH, and 3(8%) edema (13). Another study on patients undergoing shunt surgery due to hydrocephalus following post-traumatic injury showed ICH including IVH in 34 patients, traumatic SAH in 14 patients, acute SDH in 5 patients, EDH along with skull fracture in 4 patients, and skull fracture in 3 patients [19]. Another study, based on CT scan findings in early stages of injury, demonstrated diffuse edema in 7 patients, brain contusion in 6 patients, SAH in 3 patients, and normal CT scans in 4 individuals [21].
Studies have also found that the incidence of hydrocephalus is higher when injuries due to intracerebral trauma such as brain contusion, and intracerebral edema and hematoma occur [19]. For example, intracerebral hematoma via mass effect can cause blockage of CSF flow and thus play a role in hydrocephalus [21]. In our study, 11 patients underwent surgical treatment, and only 3 patients with PTH were treated medically. In the study of Sarkari et al., 25 patients (65%) were put under decompressive craniectomy (DC) and 13 patients (65%) were treated medically [13].
In another study, lumbar-peritoneal (LP) shunts were placed in 52 patients with communicative hydrocephalus following post-traumatic injury. Ventriculoperitoneal (VP) shunts were used in 12 patients with non-communicative hydrocephalus or patients with communicative hydrocephalus with no recovery after lumbar tapping [19]. Another study showed that 24 patients with hydrocephalus underwent VP shunt, and 1 patient with acute head injury received hyperventilation for 48 hours [21]. Based on the findings of this study, 3 patients recovered. Moderate disability in 3, severe disability in 2, vegetative state in 1, and death in 5 people occurred. In another study, 30 patients with hydrocephalus (78%) recovered clinically after VP shunting. 2 patients (5%) manifested no improvement, and 6 patients (15%) died [13].
Mazzini and colleagues in their study on patients undergoing shunt surgery found that only 1 patient recovered completely one year after traumatic injury (GOS=5), 3 patients (23%) had mild disability (GOS=4), 7 patients (54%) suffered from severe disability (GOS=3), and 2 patients (15%) in PVS [20]. Another study demonstrated that all patients recovered after surgery. 1 patient was treated with hyperventilation and was discharged after 2 weeks [21]. According to the results of our study and the low GCS means that the patient has suffered from severe brain damage and a more severe brain injury can lead to a more severe disorder in the absorption of CSF and contribute to ventricular expansion [16]. In this study, the results of the analysis regarding cranial damages accompanying hydrocephalus showed that only ICH (P=0.045) and IVH (P=0.013) had a statistically significant relationship with incidence of hydrocephalus. In the study by Tian et al., the results of X2 and Fisher exact tests demonstrated that there was a significant relationship between hydrocephalic incidence and GCS on admission [16]. A study conducted by Mazzini et al. found no relationship between the severity of hydrocephalus and the type of damage assessed by primary CT scan [20].
5. Conclusion
Hydrocephalus is one of the complications that can occur in people with trauma. According to the results of our study, this complication was more common in young patients (younger than 40 years of age) with trauma, thus it is necessary to use preventive programs to minimize the occurrence of trauma in this age group. Also, given this complication was observed in patients with mild head injury, more attention is needed in order to detect hydrocephalus, if any, as soon as possible, and these patients be treated appropriately, and hospital costs and long-term hospitalization due to lack of timely diagnosis and treatment be reduced. It is also necessary to carry out more studies such as clinical trials or cohorts in order to investigate, to a large extent, the occurrence of this complication and its causes in patients.
During the follow-up, the patients who died or became unavailable were excluded from the study. Since mortality and morbidity are observed more often in severe TBI, more patients with severe TBI were excluded compared to the patients with moderate or mild head injuries.
Acknowledgments
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Conflict of Interest
The authors declare that they have no conflicts of interest
References
Full Text: (2816 Views)
1. Introduction
Trauma is the most common cause of death in the first four decades of life [1]. In Iran, cardiovascular diseases, followed by trauma are the most frequent causes of death in all age groups [1]. Post-Traumatic Hydrocephalus (PTH) is an important and treatable complication following Traumatic Brain Injury (TBI) [2]. Various studies have reported a wide range of incidence (0 to 45%) for PTH [3]. A large part of these differences in the occurrence of PTH is due to application of different sets of clinical criteria in its diagnosis [4, 5]. PTH is not a single clinical concept, but a pathophysiologic condition with many aspects [6], which can be caused by excess production of Cerebrospinal Fluid (CSF), obstruction of normal flow of CSF or its inadequate absorption, which accumulates extreme pericerebral CSF [7]. If untreated, it can cause disability and death [7, 8].
Hydrocephalus is classified into communicative and obstructive types [9]. Obstructive hydrocephalus is the result of blockage of CSF flow within the ventricles [9]. The sylvian aqueduct is the most common position of intraventricular obstruction of CSF [10]. In communicative hydrocephalus, there is a connection between the ventricles and the lumbar CSF, despite of blocked flow of CSF at the brain surface or impaired CSF absorption [9]. In most cases, hydrocephalus occurring due to trauma is delayed communicative one. Obstructive hydrocephalus following head injury occurs rarely and is often associated with Intraventricular Hemorrhage (IVH) [11]. Some studies have reported the onset of PTH from two weeks to one year after TBI [12], and the earliest reported time for PTH incidence was seven hours [13, 14]. CT scan and MRI and other complementary techniques allow observation of the evolution and progress of hydrocephalus from the first to the last stages [15].
However, progressive ventricular expansion is observed in most patients and shunt surgery is recommended [16, 17]. The data from the studies show that if the ventricular expansion caused by PTH is not treated in a timely manner, it can lead to brain damage that may not be fully recovered by surgical procedures that change in the flow path of CSF [14]. Considering the need for timely diagnosis and treatment for patients with PTH, further studies on this complication may be effective in reducing mortality and disability and helping to improve these patients, therefore we decided to study the patients with head trauma in Rasht Poursina hospital.
2. Methods and Materials/Patients
In this study, patients with severe or moderate injury (GCS 12) and patients with mild head injury (GCS=13-15) (accompanying brain injuries observed in CT scan when the patients were admitted) entered the study. The patients who died early in the follow-up period or became unavailable or had hydrocephalus prior to head trauma or had variables with an independent role in hydrocephalus incidence were excluded from the study. Primary information including age, sex, GCS and trauma mechanism (crashes, falls, quarrel, etc.) was collected from the patients and recorded. The patients with hydrocephalus observed in their CT scan follow up were further investigated. Also, information about the type of treatment used for these patients (medical or surgical treatment) as well as the patients’ therapeutic outcomes were determined based on the Glasgow Outcome Scale (GOS) and related information was recorded. The data were entered into SPSS software (version 18) and analyzed by Fisher’s exact test and independent t-test.
3. Results
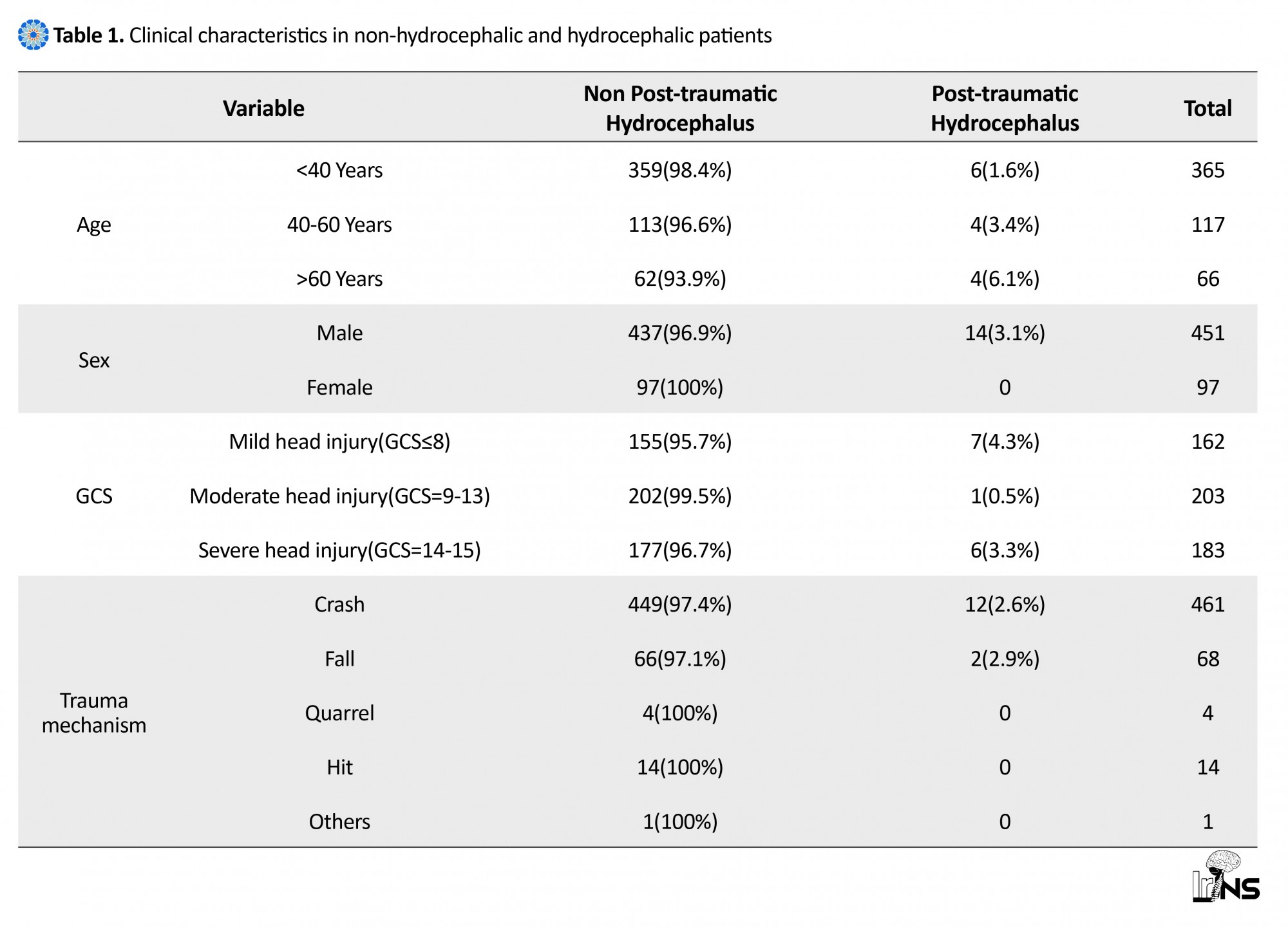
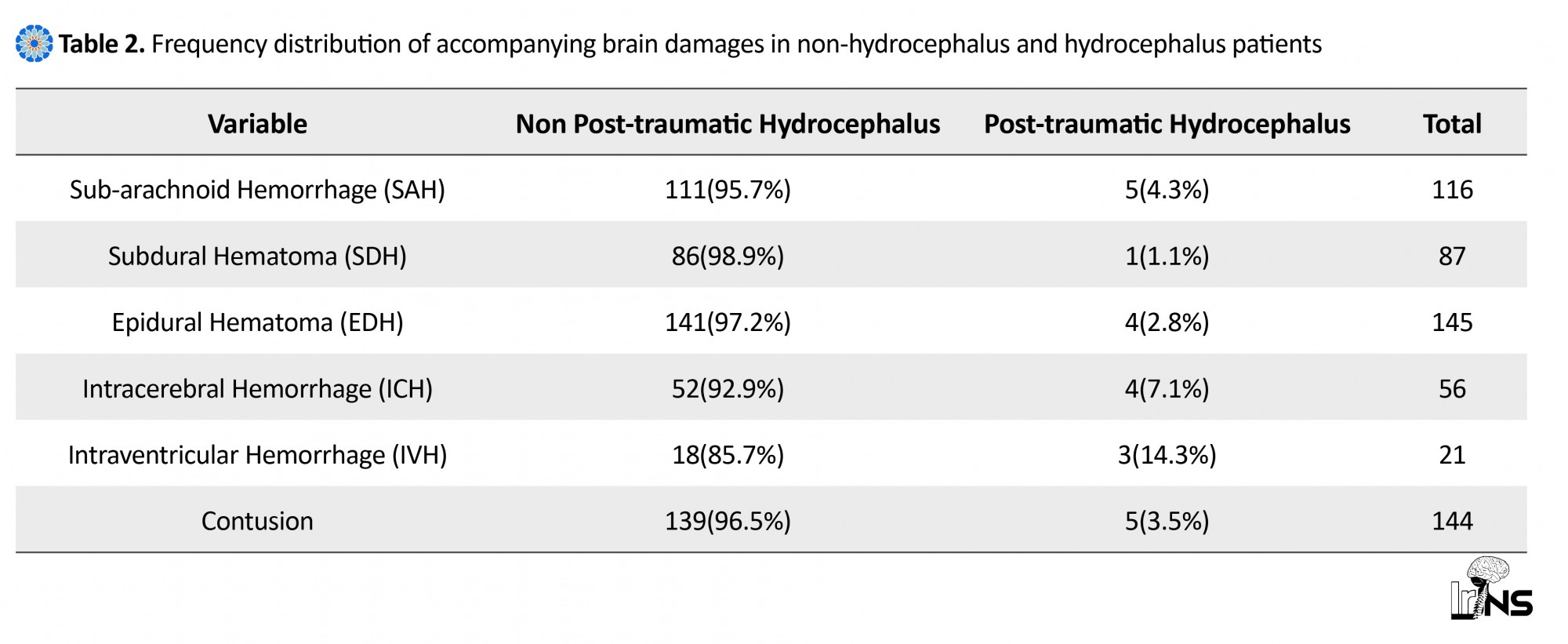
Of 548 patients with head injury examined in this study, 2.6% (14 patients) had hydrocephalus. The mean age of the patients was 44.07±24.48 years, ranging from 7 to 83 years old, mostly less than 40 years of age. All hydrocephalus patients were male. Considering the mechanism of trauma in the studied patients, two leading causes of accidents were identified as crashes (85.7%, n=12), and fall (14.3%, n=2) in patients with hydrocephalus. In this study, the level of patients’ consciousness was evaluated on the basis of GCS on admission. Accordingly, the severity of head injury was mild in 50% of patients with hydrocephalus (n=7) (GCS=13-15), and was moderate in 42.9% of the patients (n=6) (GCS=9-12), and was severe in 7.1% of the patients (only one patient) (GCS=3-8) (Table 1). The most common cranial damages observed in CT scan images in patients with hydrocephalus were Subarachnoid Hemorrhage (SAH) (35.7%, n=5) and then Epidural Hematoma (EDH) (28.6%, n=4) and Intracerebral Hemorrhage (ICH) (28.6%, n=4). 78.6% (n=11) of patients underwent surgery, and only 21.4% (n=3) of the patients with PTH were treated medically (Table 2).
Trauma is the most common cause of death in the first four decades of life [1]. In Iran, cardiovascular diseases, followed by trauma are the most frequent causes of death in all age groups [1]. Post-Traumatic Hydrocephalus (PTH) is an important and treatable complication following Traumatic Brain Injury (TBI) [2]. Various studies have reported a wide range of incidence (0 to 45%) for PTH [3]. A large part of these differences in the occurrence of PTH is due to application of different sets of clinical criteria in its diagnosis [4, 5]. PTH is not a single clinical concept, but a pathophysiologic condition with many aspects [6], which can be caused by excess production of Cerebrospinal Fluid (CSF), obstruction of normal flow of CSF or its inadequate absorption, which accumulates extreme pericerebral CSF [7]. If untreated, it can cause disability and death [7, 8].
Hydrocephalus is classified into communicative and obstructive types [9]. Obstructive hydrocephalus is the result of blockage of CSF flow within the ventricles [9]. The sylvian aqueduct is the most common position of intraventricular obstruction of CSF [10]. In communicative hydrocephalus, there is a connection between the ventricles and the lumbar CSF, despite of blocked flow of CSF at the brain surface or impaired CSF absorption [9]. In most cases, hydrocephalus occurring due to trauma is delayed communicative one. Obstructive hydrocephalus following head injury occurs rarely and is often associated with Intraventricular Hemorrhage (IVH) [11]. Some studies have reported the onset of PTH from two weeks to one year after TBI [12], and the earliest reported time for PTH incidence was seven hours [13, 14]. CT scan and MRI and other complementary techniques allow observation of the evolution and progress of hydrocephalus from the first to the last stages [15].
However, progressive ventricular expansion is observed in most patients and shunt surgery is recommended [16, 17]. The data from the studies show that if the ventricular expansion caused by PTH is not treated in a timely manner, it can lead to brain damage that may not be fully recovered by surgical procedures that change in the flow path of CSF [14]. Considering the need for timely diagnosis and treatment for patients with PTH, further studies on this complication may be effective in reducing mortality and disability and helping to improve these patients, therefore we decided to study the patients with head trauma in Rasht Poursina hospital.
2. Methods and Materials/Patients
In this study, patients with severe or moderate injury (GCS 12) and patients with mild head injury (GCS=13-15) (accompanying brain injuries observed in CT scan when the patients were admitted) entered the study. The patients who died early in the follow-up period or became unavailable or had hydrocephalus prior to head trauma or had variables with an independent role in hydrocephalus incidence were excluded from the study. Primary information including age, sex, GCS and trauma mechanism (crashes, falls, quarrel, etc.) was collected from the patients and recorded. The patients with hydrocephalus observed in their CT scan follow up were further investigated. Also, information about the type of treatment used for these patients (medical or surgical treatment) as well as the patients’ therapeutic outcomes were determined based on the Glasgow Outcome Scale (GOS) and related information was recorded. The data were entered into SPSS software (version 18) and analyzed by Fisher’s exact test and independent t-test.
3. Results
Of 548 patients with head injury examined in this study, 2.6% (14 patients) had hydrocephalus. The mean age of the patients was 44.07±24.48 years, ranging from 7 to 83 years old, mostly less than 40 years of age. All hydrocephalus patients were male. Considering the mechanism of trauma in the studied patients, two leading causes of accidents were identified as crashes (85.7%, n=12), and fall (14.3%, n=2) in patients with hydrocephalus. In this study, the level of patients’ consciousness was evaluated on the basis of GCS on admission. Accordingly, the severity of head injury was mild in 50% of patients with hydrocephalus (n=7) (GCS=13-15), and was moderate in 42.9% of the patients (n=6) (GCS=9-12), and was severe in 7.1% of the patients (only one patient) (GCS=3-8) (Table 1). The most common cranial damages observed in CT scan images in patients with hydrocephalus were Subarachnoid Hemorrhage (SAH) (35.7%, n=5) and then Epidural Hematoma (EDH) (28.6%, n=4) and Intracerebral Hemorrhage (ICH) (28.6%, n=4). 78.6% (n=11) of patients underwent surgery, and only 21.4% (n=3) of the patients with PTH were treated medically (Table 2).
The outcome of patients was evaluated based on the GOS and categorized in five groups of complete recovery, mild disability, moderate disability, severe disability, vegetative state, and death. Accordingly, 21.4% (n=3) had complete recovery. Moderate disability was found in 21.4% (n=3), severe disability in 14.3% (n=2), vegetative state in 7.14% (n=1), and death in 35.7% (n=5). Independent T-test demonstrated that no statistically significant relationship was observed between age and hydrocephalus (P=0.06), but there was a statistically significant relationship between GCS and hydrocephalus (P=0.03). Also, a statistically significant relationship was found between GCS and hydrocephalus (P=0.063) and the mechanism of trauma and hydrocephalus (P=0.817) using Fisher’s exact test showed that no statistically significant relationship was demonstrated between these variables and hydrocephalus incidence. In addition, the results of the analysis of the test about the association between accompanying cranial damages and hydrocephalus found that only ICH (P=0.045) and IVH (P=0.013) on admission had a statistically significant relationship with hydrocephalus incidence.
4. Discussion
Ventricular dilatation is a common complication in patients with TBI that can be seen in subsequent examinations on the patient and can become PTH [18]. If not diagnosed and treated, it can negatively affect the patient’s recovery and outcome [2]. Based on the results of our study, the frequency of hydrocephalus in 548 patients with head injury was obtained 14 patients (2.6%). In a study by Choi et al., 28 out of 693 (4%) patients with trauma were found to have hydrocephalus [4]. The study results of Kim et al. examining 789 patients with head injury showed that hydrocephalus was detected in 129 patients [19]. In a study on 140 patients with severe TBI, Mazzini and colleagues found that 45% of 140 patients manifested degrees of hydrocephalus, but severe hydrocephalus was found in only 11% of patients [20].
In another study by Tian et al., 36 out of 301 patients (11.96%) suffered from hydrocephalus during a three month period [16]. The mean age of the patients in this study was 44.07±24.48 years old, ranging from 7 to 83 years old. The mean age of the patients was 33.7±16 years, with the range of 2 to 65 years, in the study by Sarkari et al. [13]. In another study, the age range of patients was 2 to 89 years, and the mean age was 49.7±10.5 years [19]. In another study, the mean age of patients was 36±16 with the age range of 15 to 79 years old [20]. Bhatoe and his colleague reported the age range of patients 12 to 65 years [21].
In this study, 31.1% of men (n=14) suffered from hydrocephalus, while women showed no case of hydrocephalus. Other studies demonstrated that men had more hydrocephalus cases compared to women [13, 21]. Crashes (85.7%) and fall (14.3%) were identified as the two leading traumatic causes of hydrocephalus in patients. In another study, road accidents (68%) and then height fall (32%) were reported as the most common causes of injury [13]. In this study, head injury severity was found to be mild in most patients with hydrocephalus (n=7) (GCS=13-15) and then moderate (n=6) (GCS=9-12), and severe (n=1) (GCS=3-8). In another study, most patients (68%) had severe head injuries on admission (GCS less than or equal to 8) [13].
In a study by Kim et al., GCS on admission was categorized in four groups; less than 7 in 39 patients, between 8 and 10 in 13 patients, between 11 to 13 in 9 patients, and between 14 and 15 in 3 patients. Hydrocephalus occurred significantly higher in patients with low GCS on admission [19]. Another study showed that the mean GCS on admission in hydrocephalic patients was 13±2.8, ranging from 6 to 15 [20]. In another study, 25 patients with hydrocephalus caused by post-traumatic injury were examined. It was found that 20 patients had closed head injury. In these patients, the primary head injury in 15 patients was severe (GCS≤8) [21]. In our study, the most frequent accompanying cranial injuries observed in CT scan images were SAH (n=5), then EDH (n=4), and ICH (n=4). In another study on patients with hydrocephalus, 28 patients (76%) suffered from brain contusion, 19(50%) SAH, 18(47%) SDH, 3(8%) IVH, and 3(8%) edema (13). Another study on patients undergoing shunt surgery due to hydrocephalus following post-traumatic injury showed ICH including IVH in 34 patients, traumatic SAH in 14 patients, acute SDH in 5 patients, EDH along with skull fracture in 4 patients, and skull fracture in 3 patients [19]. Another study, based on CT scan findings in early stages of injury, demonstrated diffuse edema in 7 patients, brain contusion in 6 patients, SAH in 3 patients, and normal CT scans in 4 individuals [21].
Studies have also found that the incidence of hydrocephalus is higher when injuries due to intracerebral trauma such as brain contusion, and intracerebral edema and hematoma occur [19]. For example, intracerebral hematoma via mass effect can cause blockage of CSF flow and thus play a role in hydrocephalus [21]. In our study, 11 patients underwent surgical treatment, and only 3 patients with PTH were treated medically. In the study of Sarkari et al., 25 patients (65%) were put under decompressive craniectomy (DC) and 13 patients (65%) were treated medically [13].
In another study, lumbar-peritoneal (LP) shunts were placed in 52 patients with communicative hydrocephalus following post-traumatic injury. Ventriculoperitoneal (VP) shunts were used in 12 patients with non-communicative hydrocephalus or patients with communicative hydrocephalus with no recovery after lumbar tapping [19]. Another study showed that 24 patients with hydrocephalus underwent VP shunt, and 1 patient with acute head injury received hyperventilation for 48 hours [21]. Based on the findings of this study, 3 patients recovered. Moderate disability in 3, severe disability in 2, vegetative state in 1, and death in 5 people occurred. In another study, 30 patients with hydrocephalus (78%) recovered clinically after VP shunting. 2 patients (5%) manifested no improvement, and 6 patients (15%) died [13].
Mazzini and colleagues in their study on patients undergoing shunt surgery found that only 1 patient recovered completely one year after traumatic injury (GOS=5), 3 patients (23%) had mild disability (GOS=4), 7 patients (54%) suffered from severe disability (GOS=3), and 2 patients (15%) in PVS [20]. Another study demonstrated that all patients recovered after surgery. 1 patient was treated with hyperventilation and was discharged after 2 weeks [21]. According to the results of our study and the low GCS means that the patient has suffered from severe brain damage and a more severe brain injury can lead to a more severe disorder in the absorption of CSF and contribute to ventricular expansion [16]. In this study, the results of the analysis regarding cranial damages accompanying hydrocephalus showed that only ICH (P=0.045) and IVH (P=0.013) had a statistically significant relationship with incidence of hydrocephalus. In the study by Tian et al., the results of X2 and Fisher exact tests demonstrated that there was a significant relationship between hydrocephalic incidence and GCS on admission [16]. A study conducted by Mazzini et al. found no relationship between the severity of hydrocephalus and the type of damage assessed by primary CT scan [20].
5. Conclusion
Hydrocephalus is one of the complications that can occur in people with trauma. According to the results of our study, this complication was more common in young patients (younger than 40 years of age) with trauma, thus it is necessary to use preventive programs to minimize the occurrence of trauma in this age group. Also, given this complication was observed in patients with mild head injury, more attention is needed in order to detect hydrocephalus, if any, as soon as possible, and these patients be treated appropriately, and hospital costs and long-term hospitalization due to lack of timely diagnosis and treatment be reduced. It is also necessary to carry out more studies such as clinical trials or cohorts in order to investigate, to a large extent, the occurrence of this complication and its causes in patients.
During the follow-up, the patients who died or became unavailable were excluded from the study. Since mortality and morbidity are observed more often in severe TBI, more patients with severe TBI were excluded compared to the patients with moderate or mild head injuries.
Acknowledgments
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Conflict of Interest
The authors declare that they have no conflicts of interest
References
- alimi J, Nassaji Zavareh M, Khaji A. [Trauma mortality in six university hospitals: Tehran, IRAN (Persian)]. Tehran University Medical Journal. 2008; 65(14):22-5.
- Weintraub AH, Gerber DJ, Kowalski RG. Posttraumatic hydrocephalus as a confounding influence on brain injury rehabilitation: incidence, clinical characteristics, and outcomes. Archives of Physical Medicine and Rehabilitation. 2017; 98(2):312-9. doi: 10.1016/j.apmr.2016.08.478
- Ding J, Guo Y, Tian H. The influence of decompressive craniectomy on the development of hydrocephalus: a review. Arquivos de Neuro-Psiquiatria. 2014; 72(9):715-20. doi: 10.1590/0004-282X20140106
- Choi I, Park HK, Chang JC, Cho SJ, Choi SK, Byun BJ. Clinical factors for the development of posttraumatic hydrocephalus after decompressive craniectomy. Journal of Korean Neurosurgical Society. 2008; 43(5):227-31. doi: 10.3340/jkns.2008.43.5.227
- Cardoso ER, Galbraith S. Posttraumatic hydrocephalus—a retrospective review. Surgical Neurology. 1985; 23(3):261-64. doi: 10.1016/0090-3019(85)90092-8
- Oi S. Classification of hydrocephalus: critical analysis of classification categories and advantages of “Multi-categorical Hydrocephalus Classification” (Mc HC). Child's Nervous System. 2011; 27(10):1523-33. doi: 10.1007/s00381-011-1542-6
- Katz RT, Brander V, Sahgal V. Update on the diagnosis and management of posttraumatic hydrocephalus. American Journal of Physical Medicine & Rehabilitation. 1989; 68(2):91-6. doi: 10.1097/00002060-198904000-00009
- Kralick F, Oh J, Medina T, Noh H.. Micro-fabricated shunt to mimic arachnoid granulations for the treatment of communicating hydrocephalus. Neurochirurgica Supplementum. 2012; 114:239-42. doi: 10.1007/978-3-7091-0956-4_47
- Schapira HV. Neurology and clinical Neuroscience. New York: Elsevier; 2007.
- Cinalli G, Spennato P, Nastro A, Aliberti F, Trischitta V, Ruggiero C, et al. Hydrocephalus in aqueductal stenosis. Child’s Nervous System. 2011; 27(10):1621–42. doi: 10.1007/s00381-011-1546-2
- Fleischer AS, Huhn SL, Meislin H. Post-traumatic acute obstructive hydrocephalus. Annals of Emergency Medicine. 1988; 17(2):165–7. doi: 10.1016/s0196-0644(88)80305-6
- Iencean STM, Ianovici N, Ciurea AV. Intracranial pressure monitoring study in severe traumatic brain injury and post-traumatic hydrocephalus. Romanian Neurosurgery. 2009; 16(2):17-9.
- Sarkari A, Gupta DK, Sinha S, et al. Post-traumatic hydrocephalus : Presentation, management and outcome – An apex trauma center experience. Indian Journal of Neurotrauma. 2010; 7(2):135-8. doi: 10.1016/s0973-0508(10)80028-0
- Rodrigues D, Sharma RR, Sousa J. Post-traumatic hydrocephalus in severe head injury series of 22 cases. Pan Arab Journal of Neurosurgery. 2000; 4(2):1-10.
- Mori K. Current concept of hydrocephalus: ٍvolution of classification. Child's Nervous System. 1995; 11(9):523-31. doi: 10.1007/bf00822842
- Tian HL, Xu T, Hu J, Cui Y, Chen H, Zhou LF. Risk factors related to hydrocephalus after traumatic subarachnoid hemorrhage. Surgical Neurology. 2008; 69(3):241–6. doi: 10.1016/j.surneu.2007.02.032
- Gupta SK, Sharma T. Acute post-traumatic hydrocephalus in an infant due to aqueductal obstruction by a blood clot: a case report. Child's Nervous System. 2009; 25(3):373-6. doi: 10.1007/s00381-008-0750-1
- Zhao J, Chen Z, Xi G, Keep RF, Hua Y. Deferoxamine attenuates acute hydrocephalus after traumatic brain injury in rats. Translational Stroke Research. 2014; 5(5):586-94. doi: 10.1007/s12975-014-0353-y
- Kim SW, Lee SM, Shin H. Clinical analysis of posttraumatic hydrocephalus. Journal of Korean Neurosurgical Society. 2005; 38(3):211-4.
- Mazzini L, Campini R, Angelino E, Rognone F, Pastore I, Oliveri G. Posttraumatic hydrocephalus: a clinical , neuroradiologic, and neuropsychologic assessment of long-term outcome. Archives of Physical Medicine and Rehabilitation. 2003; 84(11):1637-41. PMID: 14639563
- Bhatoe HS, Batish VK. Post head injury hydrocephalus. Indian Journal of Neurotrauma. 2005; 2(2):131-3. doi: 10.1016/s0973-0508(05)80028-0
Type of Study: Research |
References
1. alimi J, Nassaji Zavareh M, Khaji A. [Trauma mortality in six university hospitals: Tehran, IRAN (Persian)]. Tehran University Medical Journal. 2008; 65(14):22-5.
2. Weintraub AH, Gerber DJ, Kowalski RG. Posttraumatic hydrocephalus as a confounding influence on brain injury rehabilitation: incidence, clinical characteristics, and outcomes. Archives of Physical Medicine and Rehabilitation. 2017; 98(2):312-9. doi: 10.1016/j.apmr.2016.08.478 [DOI:10.1016/j.apmr.2016.08.478]
3. Ding J, Guo Y, Tian H. The influence of decompressive craniectomy on the development of hydrocephalus: a review. Arquivos de Neuro-Psiquiatria. 2014; 72(9):715-20. doi: 10.1590/0004-282X20140106 [DOI:10.1590/0004-282X20140106]
4. Choi I, Park HK, Chang JC, Cho SJ, Choi SK, Byun BJ. Clinical factors for the development of posttraumatic hydrocephalus after decompressive craniectomy. Journal of Korean Neurosurgical Society. 2008; 43(5):227-31. doi: 10.3340/jkns.2008.43.5.227 [DOI:10.3340/jkns.2008.43.5.227]
5. Cardoso ER, Galbraith S. Posttraumatic hydrocephalus—a retrospective review. Surgical Neurology. 1985; 23(3):261-64. doi: 10.1016/0090-3019(85)90092-8 [DOI:10.1016/0090-3019(85)90092-8]
6. Oi S. Classification of hydrocephalus: critical analysis of classification categories and advantages of "Multi-categorical Hydrocephalus Classification" (Mc HC). Child's Nervous System. 2011; 27(10):1523-33. doi: 10.1007/s00381-011-1542-6 [DOI:10.1007/s00381-011-1542-6]
7. Katz RT, Brander V, Sahgal V. Update on the diagnosis and management of posttraumatic hydrocephalus. American Journal of Physical Medicine & Rehabilitation. 1989; 68(2):91-6. doi: 10.1097/00002060-198904000-00009 [DOI:10.1097/00002060-198904000-00009]
8. Kralick F, Oh J, Medina T, Noh H.. Micro-fabricated shunt to mimic arachnoid granulations for the treatment of communicating hydrocephalus. Neurochirurgica Supplementum. 2012; 114:239-42. doi: 10.1007/978-3-7091-0956-4_47 [DOI:10.1007/978-3-7091-0956-4_47]
9. Schapira HV. Neurology and clinical Neuroscience. New York: Elsevier; 2007.
10. Cinalli G, Spennato P, Nastro A, Aliberti F, Trischitta V, Ruggiero C, et al. Hydrocephalus in aqueductal stenosis. Child's Nervous System. 2011; 27(10):1621–42. doi: 10.1007/s00381-011-1546-2 [DOI:10.1007/s00381-011-1546-2]
11. Fleischer AS, Huhn SL, Meislin H. Post-traumatic acute obstructive hydrocephalus. Annals of Emergency Medicine. 1988; 17(2):165–7. doi: 10.1016/s0196-0644(88)80305-6 [DOI:10.1016/S0196-0644(88)80305-6]
12. Iencean STM, Ianovici N, Ciurea AV. Intracranial pressure monitoring study in severe traumatic brain injury and post-traumatic hydrocephalus. Romanian Neurosurgery. 2009; 16(2):17-9.
13. Sarkari A, Gupta DK, Sinha S, et al. Post-traumatic hydrocephalus : Presentation, management and outcome – An apex trauma center experience. Indian Journal of Neurotrauma. 2010; 7(2):135-8. doi: 10.1016/s0973-0508(10)80028-0 [DOI:10.1016/S0973-0508(10)80028-0]
14. Rodrigues D, Sharma RR, Sousa J. Post-traumatic hydrocephalus in severe head injury series of 22 cases. Pan Arab Journal of Neurosurgery. 2000; 4(2):1-10.
15. Mori K. Current concept of hydrocephalus: ٍvolution of classification. Child's Nervous System. 1995; 11(9):523-31. doi: 10.1007/bf00822842 [DOI:10.1007/BF00822842]
16. Tian HL, Xu T, Hu J, Cui Y, Chen H, Zhou LF. Risk factors related to hydrocephalus after traumatic subarachnoid hemorrhage. Surgical Neurology. 2008; 69(3):241–6. doi: 10.1016/j.surneu.2007.02.032 [DOI:10.1016/j.surneu.2007.02.032]
17. Gupta SK, Sharma T. Acute post-traumatic hydrocephalus in an infant due to aqueductal obstruction by a blood clot: a case report. Child's Nervous System. 2009; 25(3):373-6. doi: 10.1007/s00381-008-0750-1 [DOI:10.1007/s00381-008-0750-1]
18. Zhao J, Chen Z, Xi G, Keep RF, Hua Y. Deferoxamine attenuates acute hydrocephalus after traumatic brain injury in rats. Translational Stroke Research. 2014; 5(5):586-94. doi: 10.1007/s12975-014-0353-y [DOI:10.1007/s12975-014-0353-y]
19. Kim SW, Lee SM, Shin H. Clinical analysis of posttraumatic hydrocephalus. Journal of Korean Neurosurgical Society. 2005; 38(3):211-4.
20. Mazzini L, Campini R, Angelino E, Rognone F, Pastore I, Oliveri G. Posttraumatic hydrocephalus: a clinical, neuroradiologic, and neuropsychologic assessment of long-term outcome. Archives of Physical Medicine and Rehabilitation. 2003; 84(11):1637-41. PMID: 14639563 [DOI:10.1053/S0003-9993(03)00314-9]
21. Bhatoe HS, Batish VK. Post head injury hydrocephalus. Indian Journal of Neurotrauma. 2005; 2(2):131-3. doi: 10.1016/s0973-0508(05)80028-0 [DOI:10.1016/S0973-0508(05)80028-0]
Send email to the article author
| Rights and Permissions | |
 |
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License. |